Atypical optic neuritis resulting in a diagnosis of MOGAD

Abstract
Background
The most common systemic cause of optic neuritis continues to be multiple sclerosis.1 However, within the past decade other less common causes have been identified, such as neuromyelitis optica spectrum disorder and myelin oligodendrocyte antibody-associated disease. Previously, patients presenting with these conditions were likely grouped into having multiple sclerosis. Today, thankfully, advancements in laboratory testing and increased knowledge of the central nervous system have allowed them to be identified as distinct nosological entities. Identifying the cause of optic neuritis has many important implications, namely the ability to give an accurate prognosis, both visually and systemically.
Case Report
A 31-year-old male presented to the eye clinic for an urgent exam and reported acute, worsening vision loss in the left eye with pain around the eye as well as on eye movements. He also reported many systemic symptoms around the onset of vision loss, such as diarrhea, body aches, fatigue, and dizziness. Dilated fundus examination revealed severe disc edema of the left eye. The patient was then referred to the emergency room for magnetic resonance imaging and laboratory testing. After determining the cause to be non-infectious, the patient was treated with high-dose intravenous corticosteroids. There was a dramatic improvement in his visual acuity within a day of treatment initiation. Later, the laboratory tests were received and suggested a diagnosis of myelin oligodendrocyte antibody-associated disease.
Conclusion
The following case demonstrates an atypical presentation of optic neuritis and many factors that pointed in the direction of a cause different than multiple sclerosis. Given the rarity of neuromyelitis optica spectrum disorder and myelin oligodendrocyte antibody-associated disease, laboratory testing should be performed in cases with a higher pretest probability to avoid excessive false positives.
Keywords: optic neuritis, myelin oligodendrocyte glycoprotein antibodies, myelin oligodendrocyte glycoprotein antibody disease, multiple sclerosis, ophthalmology.
Introduction
Over the past few decades, a diagnosis of optic neuritis has almost become almost synonymous with multiple sclerosis in the minds of many clinicians. While multiple sclerosis continues to be the most common cause of optic neuritis, improvements in technology and laboratory testing have uncovered other demyelinating conditions as causes of optic neuritis.1 Neuromyelitis optica spectrum disorder (NMOSD) and myelin oligodendrocyte antibody-associated disease (MOGAD) are two distinct nosological entities recently discovered and can present with optic neuritis. Radiologic and laboratory testing are available to aid in a diagnosis; however, clinicians should be aware of when to suspect these less common causes and determine the appropriateness of ordering testing.
Case Presentation
G.D. is a 31-year-old Caucasian male who presented as an established patient for an urgent eye exam due to sudden onset vision loss in the left eye that had gradually worsened over two weeks. His symptoms were accompanied by severe pain on eye movements. He also noted several systemic symptoms that began a week prior to onset, including mild left-sided headache, diarrhea, muscle aches, fatigue, and dizziness. He denied having a fever.
His prior ocular history was unremarkable except for mild myopic astigmatism. His systemic history included depression, anxiety, and vitamin D deficiency – all managed by hydroxyzine 25 mg TID, ergocalciferol 1,250 mcg/day, and cholecalciferol 50 mcg/day.
Initial Examination
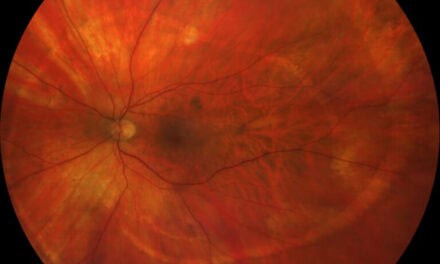
The best corrected visual acuities were 20/20 OD and 20/250 OS (pinhole no improvement). An afferent pupillary defect was noted in the left eye, as well as severe pain OS on extraocular movements despite being full range of motion OU. Intraocular pressures were 8 mmHg OD and 10 mmHg OS. Anterior segment evaluation was unremarkable, including lids, conjunctiva, cornea, iris, and anterior chamber. A dilated fundus examination with 1% tropicamide was then performed. His right eye had a healthy optic nerve with a cup-to-disc ratio of 0.20 horizontally and vertically with unremarkable macula, vasculature, and peripheral retina (see Figure 1). In the left eye, severe optic disc edema with peripapillary splinter hemorrhages and diffusely elevated retinal nerve fiber layer was noted. The macula and peripheral retina were unremarkable in the left eye (see Figure 2).

Figure 1: Image of the right eye. Healthy optic nerve with CDR of 0.20 round, normal macula, vasculature, and periphery.

Figure 2: Image of the left eye. Severe disc and RNFL edema with peripapillary intraretinal hemorrhages. Macula and peripheral retina normal.
Spectral-domain optical coherence tomography was performed on the optic nerves in both eyes (see Figures 3 and 4). The right eye’s average retinal nerve fiber layer thickness was 109 µm with overall normal thickness in all four quadrants (superior: 135 µm, nasal: 87 µm, temporal: 67 µm, and inferior: 148 µm). The left eye’s average retinal nerve fiber layer thickness was 311 µm with measurements outside of the normative database in all four quadrants (superior: 454 µm, nasal: 420 µm, temporal: 163 µm, and inferior: 207 µm). Visual field testing was deferred at this visit due to the poor visual acuity in his affected eye.

Figure 3: SD-OCT of the RNFL demonstrating significant RNFL elevation in the left eye. Right eye RNFL thickness is within normal ranges.

Figure 4: SD-OCT raster of the optic nerve of the left eye. Dramatic elevated appearance suggestive of disc edema.
The patient was instructed to report to the hospital’s emergency room immediately for bloodwork, chest X-ray, and MRI imaging of the brain and orbits. A consult to neurology was also placed at this time. Bloodwork included CBC with differential, ESR, CRP, syphilis IgG, ANA, lysozyme, ANCA, ACE, Lyme titer, B. henselae, B. quintana, neuromyelitis optica IgG (anti-aquaporin 4), anti-myelin associated glycoprotein (MAG) IgM western blot, HIV, several polymerase chain reactions for viruses (including, but not limited to, adenovirus, coronavirus, rhinovirus, enterovirus, influenza A and B, RSV), and anti-myelin oligodendrocyte glycoprotein (MOG) cell-based assay and titer. All labs were unremarkable except for elevated lysozyme (12.2 mcg/mL) and positive anti-MOG cell-based assay with a titer of >1:160. MRI imaging of brain and orbits demonstrated abnormal T2 signal and enhancement involving the intraorbital segment of the left optic nerve, consistent with optic neuritis. There was no abnormality found in the brain parenchyma. Chest X-ray was normal. The patient denied experiencing Lhermitte or Uthoff signs.
Given the findings consistent with optic neuritis and with support from neurology, G.D. was admitted to the hospital and started on intravenous methylprednisolone 1 g daily for 72 hours. The leading differential diagnoses were optic disc edema due to post-viral syndrome or MOG-antibody disease, given that the onset was accompanied by many systemic symptoms.
One Day Follow-up
G.D. was evaluated the following day while admitted to the hospital. At this time, he had been on IV methylprednisolone 1 g daily for 16 hours. G.D. reported that his vision and pain on eye movements in the left eye were improving. His visual acuities were 20/20 OD and 20/50 OS with stable pupil and posterior findings to the previous day. This assessment was done via bedside; therefore, formal visual field testing was deferred. Neurology evaluated the patient and recommended continuing with the IV methylprednisolone as planned. A lumbar puncture was also recommended, but the patient declined.
Two Day Follow-up
At the second day follow-up, G.D. reported that his vision had continued to improve and pain on eye movements was completely resolved. His visual acuities were 20/20 OD and 20/25 OS with stable pupil and posterior segment findings. This assessment was done via bedside; therefore, formal visual field testing was deferred. Intravenous methylprednisolone, 1 g daily, was continued.
Three Day Follow-up
G.D. reported that visual acuity in the left eye continued to improve, and the pain with eye movements had continued to be resolved. Visual acuities at this visit were 20/20 OD and 20/25 OS with stable pupil and posterior findings. This assessment was done via bedside; therefore, formal visual field testing was deferred. At this time, IV methylprednisolone 1 g daily was discontinued, and an oral prednisone taper over two months (120 mg X 7 days, 100 mg X 7 days, 80 mg X 7 days, 60 mg X 7 days, 40 mg X 7 days, 20 mg X 7 days, and 10 mg X 7 days) was recommended due to the growing suspicion of MOG-antibody disease over other causes. G.D. was discharged from the hospital, and a two-week follow-up appointment was scheduled.
After the three-day follow-up G.D. was lost to follow-up. Six weeks after initial presentation and testing, the anti-MOG cell-based assay came back positive with a titer of >1:160. It is uncertain if this time frame for results is typical. Given the limited number of laboratories with the ability to perform this test, the specimen was sent several states away. Perhaps this regional difference, along with the need for a titer after a positive assay, resulted in the longer period of time between obtaining the test and receiving the results. Both the patient and his primary care physician were notified, and moving forward he will be evaluated yearly by neurology.
Nearly a year later, G.D. presented to the eye clinic for his annual exam. He denied having any recurrences since his treatment. His best-corrected visual acuities were 20/20 OD and 20/20 OS. Intraocular pressures were 15 mmHg OD and 15 mmHg OS. Anterior segment evaluation, including lids, conjunctiva, cornea, iris, and anterior chamber, was unremarkable. A dilated fundus examination with 1% tropicamide was then performed. His right eye had a healthy optic nerve with a cup-to-disc ratio of 0.20 horizontally and vertically with unremarkable macula, vasculature, and peripheral retina (stable to Figure 1). The left eye had a cup-to-disc ratio of 0.25 horizontally and vertically with faint temporal pallor. There was no edema of the disc or retinal nerve fiber layer. Macula and peripheral retina were unremarkable in the left eye. Visual field testing was performed with a mean deviation of -0.01 dB (VFI 100%) in the right eye (see Figure 5) and -1.77 dB (VFI 99%) in the left eye (see Figure 6). OCT of the optic nerves showed stable findings in the right eye and thinner RNFL in the left eye, particularly temporally and superiorly (see Figure 7). G.D. is currently not on any treatment for his MOGAD. He has an upcoming appointment with neurology, as he missed the initial appointment scheduled after his diagnosis. G.D. was quite pleased with the outcome and described no difference between the eyes.

Figure 5: 30-2 Visual field testing of the right eye at one-year follow-up. Mean deviation: -0.01 dB; VFI: 100%.

Figure 6: 30-2 Visual field testing of the left eye at one-year follow-up. Mean deviation: -1.77 dB; VFI: 99%.

Figure 7: SD-OCT of the RNFL. Notice the left eye has diffusely thinner RNFL compared to the right eye, particularly temporally and superiorly—right eye RNFL thickness within normal ranges.
Discussion
Over the past few decades, there has been a substantial increase in the understanding of optic neuritis, beginning with the Optic Neuritis Treatment Trial (ONTT).2 While the most common chronic inflammatory demyelinating disease of the central nervous system continues to be multiple sclerosis (MS), the discovery and use of new biomarkers have created distinctions between MS-related optic neuritis and other less common causes of optic neuritis such as neuromyelitis optica spectrum disorder (NMOSD) and myelin oligodendrocyte glycoprotein antibody disease (MOGAD). The emphasis of this discussion will be on MOGAD, as it is the leading differential diagnosis.
Pathophysiology
Myelin oligodendrocyte glycoprotein (MOG) is located on the myelin surface and is found exclusively in the central nervous system. Although its exact purpose is unclear, MOG may play a role in regulating oligodendrocyte microtubule stability and mediating the complement cascade. Compared to other glycoproteins, MOG is only found in small amounts within myelin, but its external location on myelin sheaths may make MOG more susceptible to antibodies and T-cell response.2
MOGAD is an inflammatory demyelinating disease of the central nervous system that occurs in the presence of serum MOG antibodies. It is now recognized as a distinct nosological entity separate from MS and NMOSD. Despite some clinical overlap between MS, NMO, and MOGAD, identifying which inflammatory demyelinating cause of optic neuritis has important clinical implications, specifically regarding prognosis.
Presentation of MOGAD
A literature review of MOGAD is challenging due to the recent nosological distinction of the disease. Given limited data, conclusions can be difficult to make as studies can have quite different results. Overall, several factors are consistent. MOGAD can occur in all decades of life but has a median onset of early to mid-thirties. There seems to be a very slight predilection for women, which is more prevalent in Caucasians. The presenting symptom that leads to a diagnosis of MOGAD is most commonly optic neuritis (54-95%),4,5 followed by myelitis (numbness, tingling, weakness of limbs), acute disseminated encephalomyelitis (ADEM; fever, headache, nausea/vomiting, and neurological changes including weakness in arms or legs, drowsiness, confusion, paresthesias, ataxia),5 or an ADEM-like presentation.6 Optic neuritis may present as unilateral (22.7-60.6%) or bilateral (9.1-40.4%), with initially moderate-to-severe vision loss (median of 20/60 – 20/199; 34.8% had worse than 20/200 visual acuity).7,8 Periorbital pain is a common accompanying symptom (89.4%), and disc edema is often severe and present in 79.2% of cases.7
MRI findings often include a more edematous and extended inflammatory pattern of the optic nerve, specifically the intraorbital segment (93.3%).4,7 Chiasmal and post-chiasmal involvement on MRI is rare. Brain MRI imaging can vary based on the study, ranging from 33-66% having a normal appearance of the brain at the time of presentation. Patients with abnormal brain MRIs typically showed nonspecific T2 hyperintensities, though this varies based on the study. Spinal cord MRIs in MOGAD patients were abnormal in just over half of the patients. These lesions were often short T2 hyperintensities and occurred predominantly in the cervical or thoracic region.4
A relapsing course, most commonly involving the optic nerve, is noted in patients with MOGAD and has been reported to affect 44-83% of patients.5 The impact of relapses is variable and needs more research as there are limited longitudinal studies currently. However, when compared to NMOSD, MOGAD patients tend to have better visual field outcomes despite also having frequent recurrences.
Differential Diagnoses
Some clinical overlap between MOGAD, MS and NMOSD is expected. Important differences in clinical and radiologic phenotype will assist in distinguishing between these three conditions.
MS-related optic neuritis sometimes referred to as “typical optic neuritis,” tends to have a mild predilection for women over men, with onset often in the third and fourth decades of life. The typical presentation is unilateral vision loss or disturbances, with pain on eye movements. Optic disc edema is present in approximately ⅓ of cases and is often mild when present. Often symptoms of dyschromatopsia may be more dramatic than visual acuity loss. Systemic symptoms are less common unless the patient has been diagnosed with multiple sclerosis prior to the initial episode of optic neuritis. MRI imaging of the optic nerve may demonstrate enhancement near the anterior aspect of the optic nerve. Brain MRI imaging may reveal brain abnormalities, such as periventricular white matter lesions, in addition to focal hyperintensity of the optic nerve. The presence of brain lesions consistent with demyelination increases the subsequent risk of developing MS in the future.1 Visual prognosis is good, as demonstrated in the Optic Neuritis Treatment Trial.2
NMOSD-related optic neuritis tends to have a stronger predilection for women than men, with onset often at similar ages to MS and MOGAD. Its presentation is often bilateral and simultaneous optic neuritis with severe vision loss. MRI imaging often shows longer portions of optic nerve hyperintensity that may or may not involve the optic chiasm. Brain MRI is often abnormal and with multiple lesions of the white matter, diencephalon, and medulla. Systemic symptoms may occur in association with the onset of optic neuritis. Most frequently, area postrema dysfunction (nausea/vomiting) is noted, but also longitudinally extensive transverse myelitis (LETM; back pain, paresthesias, weakness in legs that progresses upwards, bladder or bowel incontinence).11 Rarely, ADEM may occur. Visual prognosis is poor, and recurrences are common.7
Diagnosis of MOGAD
A diagnosis of MOGAD can be made by seropositivity for anti-MOG antibodies accompanied by supporting clinical and radiological presentation. An important factor to consider is the accuracy of the laboratory tests for diagnosing MOGAD, especially given its rarity. International comparative studies have demonstrated that live cell-based assays for anti-MOG IgG antibodies yield the highest specificity. However, widely testing for anti-MOG antibodies in any patient with optic neuritis will lead to increased chances for false positives.
A study from the Mayo Clinic evaluated the positive predictive value (PPV) of MOG-IgG1 testing in their tertiary referral center setting. The study included 1260 patients tested for MOG-IgG1 at the Mayo Clinic over two years, with 92 (7.3%) positive results. Of the 92 positive tests, 26 (28%) were false positives, which gives a positive predictive value of MOG-IgG1 of 72%. However, the PPV varied with autoantibody titer and clinical MRI presentation at the time of testing. For example, an autoantibody titer of >/= 1:1000 had 100% PPV, 1:100 had a PPV of 82%, and 1:20-1:40 had a PPV of 51%. Acute optic neuropathy was noted in 31 patients at onset and had a PPV of 97%, which was one of the greatest predictive factors for a true positive result (second to multifocal CNS demyelination, which had 100% PPV). Important to note that when MOG-IgG1 testing was performed in patients presenting with phenotypes atypical for MOGAD, the PPV dropped to as low as 0%. This study demonstrated that MOG-IgG1 testing can be highly specific given a higher pretest probability and with higher titers. Indiscriminately testing for MOGAD can lead to a higher likelihood of false positives, and caution is advised when interpreting low-titer positivity in cases that are atypical for MOGAD.12
Another important note is that in patients who present with atypical optic neuritis, both NMOSD and MOGAD should be considered and pursued. A consensus exists that AQP4-IgG and MOG-IgG do not coexist.9 Results that are positive for both AQP4-IgG and MOG-IgG should be considered carefully. Retesting in this scenario is strongly encouraged.
Ultimately, the reasoning behind testing G.D. for anti-MOG antibodies (as well as anti-AQP4 antibodies) was due to the severity of the optic neuritis and pain – in addition to his systemic symptoms. Initially, a post-viral cause was suspected, but all viral labs returned normal. This resulted in pursuing other, less common causes of optic neuritis. Given the positive anti-MOG IgG cell-based assay with a titer of 1:160, the severity of the optic disc edema, and MRI results showing intraorbital enhancement of the optic nerve, a confident diagnosis of MOGAD can be made. A spinal cord MRI may prove useful in cases more ambivalent in findings. In this case, the anti-MOG IgG cell-based assay results were received approximately two months later. Despite the delay, the dramatic improvement after treatment would also support the diagnosis of MOGAD over NMOSD.
Treatment and Management of MOGAD
Treatment guidelines have not been well established for MOGAD-related optic neuritis. However, due to its similarities to optic neuritis of MS and NMOSD, it is often treated with a similar approach.13 When determining appropriate treatment options for optic neuritis, it is important to consider the following goals: treating and improving the current episode, reducing pain (when present), and reducing recurrences.
A steroid pulse therapy for optic neuritis includes 1000 mg/day intravenous methylprednisolone that usually occurs for three days. Following the three days of intravenous steroids, the patient is put on oral prednisolone taper starting with 0.5 mg/kg/day. The steroid is then tapered by 5 mg every 5-7 days. When prednisolone is below 20 mg/day, azathioprine 50 mg-100 mg/day is added. These medications are continued until the prednisolone taper is complete, and then both are discontinued simultaneously.14 Additionally, some studies show that adding plasma exchange as an additional treatment outcome to the steroid pulse therapy can improve the final visual acuity recovery or be used when recurrences occur. However, due to the smaller sample sizes of studies, these trials were not randomized or had control groups.13
Regarding reduction and management of pain, there are no articles confirming adequate steroid dosing to achieve this endpoint. However, in studies specifically for NMOSD optic neuritis patients, pain is typically relieved with continuous dosing of around 20 mg/day of oral prednisolone. Unfortunately, when the steroid is tapered, pain usually returns. Therefore medications such as azathioprine and methotrexate are considered for long-term pain management and to reduce the side effects of long-term steroid usage. Research is still needed to prove that this is the most appropriate treatment strategy for patients with MOGAD optic neuritis.14
Considering appropriate treatment options and their role in preventing recurrence rates in patients with NMOSD and MOGAD optic neuritis is important. However, it can be challenging to draw conclusions on the best treatment to reduce recurrences in MOGAD patients. This is due to the small amount of evidence regarding treatment for MOGAD optic neuritis and the fact that MOGAD patients tend to have worse recurrence rates. When NMOSD patients recur, plasma exchange or blood purification therapy may be used. Therefore, this can often be considered an appropriate treatment option; however, it needs to be approached with caution due to the differences between recurrence rates and severity among NMOSD patients vs. MOGAD patients.14
Tocilizumab is a medication found to help NMOSD patients with relapsing optic neuritis. This interleukin-6 receptor blockade medication was recently studied in patients with relapsing MOGAD optic neuritis14. In this retrospective study, 11 of the 14 patients with relapsing MOGAD optic neuritis were treated with tocilizumab for >12 months and were relapse-free for two years. This study is encouraging; however, future controlled, prospective studies over a longer time are required to help define the role of tocilizumab in preventing recurrences for MOGAD patients.
Given the somewhat recent distinction of MOGAD from variants of multiple sclerosis, much is left to be discovered regarding the prognosis of the systemic and ocular complications of the disease. Perhaps the best management at this moment is diagnosing and referring these patients to a neurologist who has experience with MOGAD.
Conclusion
While the most common cause of optic neuritis continues to be multiple sclerosis, other inflammatory demyelinating conditions often present with optic neuritis as the primary onset of the disease. Clinicians should think more broadly than multiple sclerosis when considering the etiology of optic neuritis. When patients present with atypical optic neuritis – such as severe vision loss, bilateral cases, and abnormal systemic symptoms – NMOSD and MOGAD should be considered as differential diagnoses. Ordering laboratory tests (such as anti-AQP4 IgG and anti-MOG IgG) for these conditions is only appropriate in these atypical cases, as indiscriminate testing will increase the chance of receiving false positives. Additionally, a spinal cord MRI may be helpful in cases with uncertainty on the diagnosis. Overall, all three conditions are treated in a similar manner at this moment, but treatment and management may vary moving forward as more is uncovered about these conditions. Optometrists should be aware of the appropriate tests to order (bloodwork, MRI of the brain, optic nerve, and spinal cord) and pursue less common causes of optic neuritis when the presentation is atypical of MS-related optic neuritis.
Financial Disclosures
The authors do not have any relevant financial or non-financial conflicts of interest to disclose.
References
- Abel A, McClelland C, Lee MS. Critical review: Typical and atypical optic neuritis. Surv Ophthalmol. 2019 Nov-Dec;64(6):770-779. doi: 10.1016/j.survophthal.2019.06.001. Epub 2019 Jun 20. PMID: 31229520.
- Beck RW, Cleary PA, Anderson MM Jr, Keltner JL, Shults WT, Kaufman DI, et al. A randomized, controlled trial of corticosteroids in the treatment of acute optic neuritis. The Optic Neuritis Study Group. N Engl J Med. 1992 Feb 277;326(9):581-8.
- Ambrosius W, Michalak S, Kozubski W, Kalinowska A. Myelin Oligodendrocyte Glycoprotein Antibody-Associated Disease: Current Insights into the Disease Pathophysiology, Diagnosis and Management. Int J Mol Sci. 2020 Dec 24;22(1):100. doi: 10.3390/ijms22010100. PMID: 33374173; PMCID: PMC7795410.
- Ramanathan S, Prelog K, Barnes EH, Tantsis EM, Reddel SW, Henderson AP, et al. Radiological differentiation of optic neuritis with myelin oligodendrocyte glycoprotein antibodies, aquaporin-4 antibodies, and multiple sclerosis. Mult Scler. 2016 Apr;22(4):470-82. doi: 10.1177/1352458515593406. Epub 2015 Jul 10. PMID: 26163068.
- Wynford-Thomas R, Jacob A, Tomassini V. Neurological update: MOG antibody disease. J Neurol. 2019 May;266(5):1280-1286. doi: 10.1007/s00415-018-9122-2. Epub 2018 Dec 19. PMID: 30569382; PMCID: PMC6469662.
- Nationalmssociety.org [Internet]. Acute Disseminated Encephalomyeitis; c2023 [cited 2023 June 6]. Available from: https://www.nationalmssociety.org/What-is-MS/Related-Conditions/Acute-Disseminated-Encephalomyelitis-(ADEM)#section-1
- Biotti D, Bonneville F, Tournaire E, Ayrignac X, Dallière CC, Mahieu L, et al. Optic neuritis in patients with anti-MOG antibodies spectrum disorder: MRI and clinical features from a large multicentric cohort in France. J Neurol. 2017 Oct;264(10):2173-2175.
- Mariotto S, Ferrari S, Monaco S, Benedetti MD, Schanda K, Alberti D, et al. Clinical spectrum and IgG subclass analysis of anti-myelin oligodendrocyte glycoprotein antibody-associated syndromes: a multicenter study. J Neurol. 2017 Dec;264(12):2420-2430. doi: 10.1007/s00415-017-8635-4. Epub 2017 Oct 23. PMID: 29063242; PMCID: PMC5688213.
- Rarediseases.org [Internet]. Transverse Myelitis; c2023 [cited 2023 June 6]. Available from: https://rarediseases.org/rare-diseases/transverse-myelitis/
- Sechi E, Buciuc M, Pittock SJ, Chen JJ, Fryer JP, Jenkins SM, et al. Positive Predictive Value of Myelin Oligodendrocyte Glycoprotein Autoantibody Testing. JAMA Neurol. 2021 Jun 1;78(6):741-746.
- Akaishi, T., Takeshita, T., Himori, N., Takahashi, T., Misu, T., Ogawa, R., Kaneko, K., Fujimori, J., Abe, M., Ishii, T., Fujihara, K., Aoki, M., Nakazawa, T., & Nakashima, I. (2020). Rapid Administration of High-Dose Intravenous Methylprednisolone Improves Visual Outcomes After Optic Neuritis in Patients With AQP4-IgG-Positive NMOSD. Frontiers in neurology, 11, 932. https://doi.org/10.3389/fneur.2020.00932
- Kezuka, T., & Ishikawa, H. (2018). Diagnosis and treatment of anti-myelin oligodendrocyte glycoprotein antibody positive optic neuritis. Japanese journal of ophthalmology, 62(2), 101–108. https://doi.org/10.1007/s10384-018-0561-1.
- Merle H, Olindo S, Jeannin S, et al. Treatment of Optic Neuritis by Plasma Exchange (Add-On) in Neuromyelitis Optica. Arch Ophthalmol. 2012;130(7):858–862. doi:10.1001/archophthalmol.2012.1126
- Ringelstein M, Ayzenberg I, Lindenblatt G, Fischer K, et al. on behalf of the Neuromyelitis Optica Study Group (NEMOS). Interleukin-6 Receptor Blockade in Treatment-Refractory MOG-IgG–Associated Disease and Neuromyelitis Optica Spectrum Disorders Neurol Neuroimmunol Neuroinflamm Jan 2022, 9 (1) e1100; DOI: 10.1212/NXI.0000000000001100