Diagnosing Macular Telangiectasia Type 2

ABSTRACT
BACKGROUND
Macular telangiectasia (MacTel) – previously called idiopathic juxtafoveal telangiectasia – is a group of disorders that result in abnormal telangiectatic vessels in the juxtafoveal region. MacTel type 2 is the most common subtype that results in atrophic changes and sometimes subretinal neovascularization (SRN). This case report will focus on a patient with MacTel type 2 and the management and treatment options pertinent to the case.
CASE REPORT
An 89-year-old Caucasian male, previously diagnosed with moderate dry age-related macular degeneration, presented for an annual eye exam with complaints of gradually worsening vision in both eyes over the previous year. His best-corrected vision was 20/70 in the right eye and 20/200 in the left eye at distance. Fundus biomicroscopy revealed areas of hyperpigmentation and hyperreflective deposits temporal to the fovea in both eyes as well as retinal venules that appeared to course directly into the areas of hyperpigmentation. Optical coherence tomography (OCT) revealed cystic changes along with retinal pigment epithelium (RPE) disruption in both eyes. Intravenous fluorescein angiography (IVFA) revealed telangiectatic vessels surrounding the fovea temporally in the right eye while showing late retinal staining temporally in both eyes.
CONCLUSION
MacTel type 2 is a retinal disease that can lead to moderate to severe vision loss through either retinal atrophy or SRN. Its pathogenesis remains obscure and future treatment depends on continued research. This case shows how the initial diagnosis can be challenging and is often mistaken for other macular diseases. It is imperative to utilize appropriate retinal imaging and angiographic studies to determine the correct diagnosis.
Keywords: Idiopathic juxtafoveal telangiectasia, macular telangiectasia, subretinal neovascularization, age-related macular degeneration, optical coherence tomography, intravenous fluorescein angiography
INTRODUCTION
MacTel is classified into three different types. MacTel type 1 is unilateral and is characterized by the presence of microaneurysms or exudates often resulting in macular edema. MacTel type 2 is the most common and typically presents with bilateral juxtafoveal telangiectasias. The least common type, MacTel type 3 results in complete obstruction of vessels surrounding the fovea bilaterally.1-3 This case report will focus on MacTel type 2.
MacTel type 2 is an acquired progressive disease that often results in subclinical telangiectatic vessels temporal to the fovea resulting in adjacent atrophy and in some cases SRN.1-3 It is a bilateral disease but can be asymmetric with patients often presenting with unilateral complaints.2 It affects males and females equally and there is no known racial propensity. The age of onset is middle-aged and older with the mean age of 55 years old.2-3 Its prevalence is difficult to assess as most patients are asymptomatic in the early stages. However, the condition is rare, with one study suggesting the prevalence to be 0.10%.4 Up to 45% of MacTel type 2 patients have a diabetes mellitus, obesity, hypertension, or cardiovascular diseases.5 These factors, in addition to smoking, may increase the risk of clinical disease.5
Patients typically present with unilateral or bilateral gradual progressive blurred vision. Other complaints include metamorphopsia or paracentral scotomas which can be monitored with Amsler grid testing.
CASE REPORT
An 89-year-old Caucasian male presented for an annual eye exam with the complaint of gradually worsening blurry vision in both eyes over the past year. He denied metamorphopsia, diplopia, or ocular pain, and noted that the vision in his left eye was noticeably worse since his last exam one year prior. Specifically, reading had become increasingly difficult to the point where he did not think his prescription glasses helped anymore. His ocular history included type 2 diabetes mellitus without retinopathy or macular edema in either eye, moderate dry age-related macular degeneration in both eyes, pseudophakia in the right eye, and mild nuclear sclerosis in the left eye. His previously recorded best corrected visual acuities were 20/50 in the right eye and 20/70 in the left eye. His medical history was significant for type 2 diabetes mellitus for ten years, hypertension, high cholesterol, and coronary artery disease. His current medications included metformin, glipizide, lisinopril, aspirin, and AREDS 2 formula. He had no known drug allergies. His social history was negative for tobacco, alcohol, or recreational drug use. His family ocular history was negative for any eye disease. He was oriented to person, place, and time.
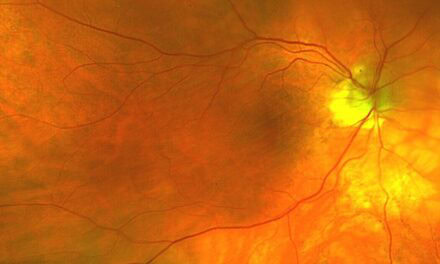
After refraction, his best corrected visual acuities were 20/70 in the right eye and 20/200 in the left eye at distance with central viewing. Entrance testing and slit lamp exam were unremarkable. Dilated exam revealed grade 1+ nuclear sclerosis with trace cortical vacuoles in the left lens. A centered posterior chamber intraocular lens without anterior or posterior capsule opacification was present in the right eye. The vitreous was clear and quiet in both eyes. Evaluation of the fundus revealed well perfused optic nerve heads with well-defined margins and a cup to disc ratio of 0.40 round in both eyes. The retinal vessels had normal caliber without hemorrhages in both eyes. The macula revealed focal areas of hyperpigmentation temporal to the fovea in both eyes with adjacent hyperreflective crystalline deposits. The foveal reflex was absent in both eyes. Of note, no drusen were present in the macula in either eye. The retinal venules in both eyes appeared to course directly into the areas of hyperpigmentation. The peripheral retina was flat and intact for all 360 degrees in both eyes.
Same day fundus photography, OCT, and IVFA imaging were performed (Figures 1-3).

Figure 1. Posterior fundus photographs demonstrating the temporal hyperpigmentation, crystalline deposits, and right-angled venules that course towards the areas of degeneration in both eyes.

Figure 2. OCT of the right eye revealed central atrophy and trace subretinal cystic changes. OCT of the left eye appeared to have extensive intraretinal and subretinal cystic changes centrally, internal limiting membrane draping, and a pigment epithelial detachment temporal to the fovea.

Figure 3. (A,B) In the arteriovenous phase of the IVFA, telangiectatic vessels are evident temporally in the right eye while dense pigment is blocking underlying dye in the left eye. (C, D) Late staining is noted temporal to the fovea in both eyes.
The patient was thoroughly educated regarding the findings and diagnosis of MacTel type 2. It was explained to the patient that there were no current treatment options available for this retinal disease. Despite the change in diagnosis, the patient wished to continue the AREDS 2 formula supplement. A referral was placed for him to be seen in the low vision clinic to help with his near vision complaints. The patient was educated to continue to monitor his vision with an Amsler grid and return to the clinic as soon as possible with any changes. The patient was scheduled to return to the clinic in 3-4 months for a dilated fundus exam and repeat OCT scans in both eyes. The patient’s follow up visit revealed stable visual acuities, fundus findings, and OCT scans in both eyes.
DISCUSSION
Macular telangiectasia type 2 patients may present with reduced visual acuity, metamorphopsia or scotoma. Visual acuities can range from 20/20 to 20/200 or worse, with severe visual impairment typically resulting from foveolar atrophy or SRN.1-3 Subretinal neovascularization develops in up to 14% of cases and occurs bilaterally in 33% of cases.6-7 In the absence of SRN, metamorphopsia was reported in 83% of eyes.8 Paracentral scotomas develop in approximately 50% of patients whose visual acuities range from 20/32 to 20/125 and 100% of patients with foveolar atrophy or SRN.9-10
Gass and Blodi classified the severity of MacTel type 2 into five stages. In stage 1, funduscopic findings may reveal subtle atrophy or graying of the parafoveal retina temporally.2-3 Unlike MacTel type 1, the parafoveal telangiectatic vessels in MacTel type 2 may initially be difficult to appreciate on fundus exam.11 As the disease progresses into stage 2, the graying of the retina may expand to surround the fovea or remain temporally.11-12 Superficial crystalline deposits may also be appreciated in the temporal quadrants and can accumulate circumferentially around the fovea.6,10-11 These deposits are refractile in nature and are found along the nerve fiber layer which may explain their absence at the horizontal raphe.13 However, the accumulation of crystalline deposits does not correlate with the severity of disease.12 Eventually the appearance of right angled venules can be seen coursing towards the temporal areas of retinal atrophy in stage 3.11,14 The foveal light reflex will be absent if the atrophic changes involve the fovea, which may mimic a macular hole.11 During stage 4, focal RPE hyperplasia along these right angled venules can be seen and in some cases migrate into the inner retina.1-3,15 Stage 5 is when SRN occurs as a result of capillary remodeling and proliferation.1-3 The SRN usually occurs temporally and near the area of RPE hyperplasia.1 Due to the difficulty in diagnosing at an early stage, utilizing the appropriate imaging such as IVFA, OCT, and fundus autofluorescence are key in making the correct diagnosis.
In cases of MacTel type 2, IVFA should be considered to provide a more definitive diagnosis. It is important to rule out other potential causes for sub-foveal cystic changes that could lead to a rapid visual decline if not treated such as exudative age-related macular degeneration and diabetic macular edema. Early in the disease course, IVFA reveals late staining temporally with foveola sparing.1 As the disease progresses, early staining of capillary walls occurs and visualization of the telangiectatic vessels become more apparent. This is followed by late retinal staining that does not enlarge or balloon.1 In advanced cases, SRN may develop in the vicinity of the retinal atrophy and rapid visual decline may ensue. This appears much like classic macular neovascularization (MNV) on IVFA, where edema or leakage of vascular contents results in enlarging or ballooning of the hyperfluorescence in the late phases.11 Unlike typical MNV seen in age-related macular degeneration, the size of the SRN in MacTel type 2 tends to be smaller.1
OCT aids in the diagnosis and is an essential part of monitoring MacTel type 2. Intraretinal cysts along the inner retina near the foveola are classic findings of MacTel type 2 on OCT scans. These cysts are not a result of leakage or exudation but rather represent tissue degradation and loss.1 As a result, a common finding characteristic to MacTel type 2 is internal limiting membrane (ILM) draping.11 This is where the thin ILM extends or “drapes” over the top of the intraretinal cyst.11 As the disease gradually progresses, these intraretinal cysts can enlarge and be seen in the outer retina as well.10 RPE hyperplasia may present over time and is seen as hyperreflective plaques that cast a shadow zone beneath them.15 In advanced cases where there is significant visual impairment, foveola atrophy and photoreceptor degradation is apparent.
Another feature of OCT is the angiography modality (OCTA) which has become very useful in the diagnosis and management of MacTel type 2. Through repeated B-scans over time it detects moving erythrocytes and can plot out a visualization of the retinal vasculature. As a result, characteristic findings such as telangiectatic vessels, right angled vessels, as well as SRN can be appreciated well before they are visible with funduscopy.11 However, OCTA is less adept at detecting leakage or staining, as compared to IVFA.16-17 Specifically in MacTel type 2, OCTA may incorrectly locate retinal vessels as the outer retina atrophies and the vessels deviate toward the RPE.16 Overall, OCTA technology remains an effective tool due to its availability, ease of use, and noninvasive qualities.
Lastly, fundus autofluorescence (FAF) photography can be a helpful tool in the diagnosis and monitoring of the disease. According to Wong et al, FAF is often the first imaging modality to show any abnormality early in the disease process when compared to IVFA, OCT, and fundus photography. Mild foveal autofluorescence is seen early on and increases as the disease progresses.18 In the more advanced stages, varied patterns of autofluorescence intensity are seen due to tissue atrophy.19
Although MacTel type 2 can lead to central vision loss, a recent study suggests that severe vision loss is uncommon. In a study by Heeren et al, a total of 4449 eyes were followed over an average span of 4.2 years. Best corrected visual acuity was 20/50 or worse in 37.3% of eyes and 20/200 or worse in 3.8%.7 More importantly, only 18.7% had bilateral acuities of 20/50 or worse and 0.7% had bilateral acuities of 20/200 or worse.7 Despite the fact that patients often have minimal decrease in binocular best corrected visual acuity, they often have significantly reduced reading ability due to central scotomas.5 Also, studies may overestimate visual acuities as the late stages of MacTel type 2 may be mistaken for other macular diseases such as age-related macular degeneration or macular dystrophies.7 Due to its association with systemic morbidities, increased mortality of patients with severe forms of MacTel type 2 may also contribute to understating the long-term visual prognosis.7
The pathogenesis of MacTel type 2 remains to be a paradox. It was originally proposed that the etiology originated from a dysfunction within the retinal vasculature.1-3 Instead, it has been postulated that the etiology results from Müller cell deterioration.10,20-21 Müller cells course radially from the internal limiting membrane to the outer nuclear layer and are an integral part in maintaining the health of the surrounding tissue as well as the retinal vascular endothelium.22 Reduction in the number of parafoveal Müller cells on histopathological analysis and lack of retinal telangiectasia detected early in the disease process on IVFA support this concept. Furthermore, the cystic changes seen on OCT are thought to be a result of tissue degeneration due to Müller cell loss rather than fluid from vascular leakage.11,20,22-23 It has also been suggested that the superficial crystalline deposits may be remnants of Müller cells due to their proximity to the internal limiting membrane.5 As the disease progresses, histopathology has revealed thickening of retinal capillary basement membranes as well as vascular endothelium and pericyte degeneration.1,5,23 Therefore, it is thought that through Müller cell degradation, the propagation of parafoveal capillary telangiectatic vessels eventually ensues. However, the exact mechanism for retinal capillary telangiectatic development remains obscure.
Treatment options remain unclear regarding MacTel type 2 without SRN. The use of anti-vascular endothelial growth factor (anti-VEGF) drugs does not appear to alter long-term outcomes. The use of anti-VEGF drugs bevacizumab and ranibizumab provided a temporary decrease in retinal thickness on OCT, however, a rebound effect was observed within 2-4 months.5 Similarly, the use of oral carbonic anhydrase inhibitors has been shown to decrease central thickness on OCT without any improvement in visual acuity in cases without SRN.24 In cases that involve SRN, bevacizumab and ranibizumab have shown to be beneficial with improvements in visual acuity, resolution of fluid, and regression of SRN.5 Due to loss of carotenoid pigment in the degenerative areas, supplementation involving lutein, meso-zeaxanthin, and zeaxanthin have been reviewed. These studies showed varied outcomes and need further large-scale investigation.25 Other modalities such as grid laser, photodynamic therapy, as well as intravitreal triamcinolone injections have not shown any visual or structural benefit.5
Due to the shift in understood pathogenesis from a vascular disorder to a neurodegenerative one, recent treatment studies have focused more on cellular fortification. Promising studies involving the use of ciliary neurotrophic factor (CNTF) are currently being reviewed. CNTF is found within humans and is a member of the cytokine family.26 It binds to certain cells and signals regulatory activities that help promote survival.26-27 In animal models with outer retinal deterioration, CNTF has been shown to reduce the amount of photoreceptor loss.25 Chew et al. conducted a study that involves the use of a semipermeable encapsulated cell implant that is attached at the pars plana.28 In this procedure, the implant, which contains modified human RPE cells programmed to produce CNTF, is released into the vitreous cavity. Ninety-nine study eyes were used and followed up at 24 months from baseline. The sham group revealed a 31% increase in retinal degeneration compared to those that received CNTF. Retinal sensitivity through microperimetry revealed a 45% greater loss in the sham group compared to the treated group. Reading speed was also reduced where it remained unchanged in the treated group. Further research is currently being conducted to determine the long-term effects and safety regarding this treatment modality.
CONCLUSION
MacTel type 2 is a retinal disease that can lead to moderate to severe vision loss through either retinal atrophy or SRN. Its pathogenesis remains obscure and future treatment depends on continued research. Ciliary neurotrophic factor appears to show promise in slowing down the disease process while anti-VEGF drugs appear to be sufficient in the treatment of SRN. Considering the degree of vision loss, low vision services should be offered to symptomatic patients. This case demonstrates how the initial diagnosis of MacTel 2 can be difficult, and that it is often mistaken for other macular diseases. It is imperative to utilize appropriate retinal imaging and angiographic studies to determine the correct diagnosis. Clinicians must be able to educate the patient on the condition as well as to recommend appropriate follow up to monitor for progression or development of SRN.
References
- Nowilaty, S. R., Al-Shamsi, H. N., & Al-Khars, W. (2010). Idiopathic juxtafoveolar retinal telangiectasis: A current review. Middle East African Journal of Ophthalmology, 17(3), 224–241.
- Gass, J. D., & Oyakawa, R. T. (1982). Idiopathic juxtafoveolar retinal telangiectasis. Archives of Ophthalmology, 100(5), 769–780.
- Yannuzzi, L. A. (2006). Idiopathic macular telangiectasia. Archives of Ophthalmology, 124(4), 450–460. https://doi.org/10.1001/archopht.124.4.450
- Klein, R., Blodi, B. A., Meuer, S. M., Myers, C. E., Chew, E. Y., & Klein, B. E. K. (2010). The prevalence of Macular Telangiectasia Type 2 in the Beaver Dam Eye Study. American Journal of Ophthalmology, 150(1).
- Kedarisetti, K. C., Narayanan, R., Stewart, M. W., Reddy Gurram, N., & Khanani, A. (2022). Macular telangiectasia type 2: A comprehensive review. Clinical Ophthalmology, Volume 16, 3297–3309.
- Charbel Issa, P., Gillies, M. C., Chew, E. Y., Bird, A. C., Heeren, T. F. C., Peto, T., Holz, F. G., & Scholl, H. P. N. (2013). Macular telangiectasia type 2. Progress in Retinal and Eye Research, 34, 49–77.
- Heeren, T. F. C., Chew, E. Y., Clemons, T., Fruttiger, M., Balaskas, K., Schwartz, R., Egan, C. A., & Charbel Issa, P. (2020). Macular telangiectasia type 2: Visual acuity, disease end stage, and the MacTel Area. Ophthalmology, 127(11), 1539–1548.
- Charbel Issa, P., Holz, F. G., & Scholl, H. P. (2009). Metamorphopsia in patients with Macular Telangiectasia Type 2. Documenta Ophthalmologica, 119(2), 133–140.
- Heeren, T. F., Clemons, T., Scholl, H. P., Bird, A. C., Holz, F. G., & Charbel Issa, P. (2015). Progression of vision loss in Macular Telangiectasia Type 2. Investigative Opthalmology & Visual Science, 56(6), 3905.
- Ledolter, A. A., Ristl, R., Palmowski‐Wolfe, A. M., Montuoro, A., Deak, G. G., Sacu, S., Holder, G. E., Schmidt‐Erfurth, U., & Ritter, M. (2021). Macular telangiectasia type 2: Multimodal assessment of retinal function and microstructure. Acta Ophthalmologica, 100(6), 1240–1252.
- Moir, J., Amin, S. V., Khanna, S., Komati, R., Shaw, L. T., Dao, D., Hariprasad, S. M., & Skondra, D. (2022). Use of OCT angiography to diagnose and manage atypical presentations of Macular Telangiectasia Type 2. International Journal of Molecular Sciences, 23(14), 7849.
- Solberg, Y., Dysli, C., Wolf, S., & Zinkernagel, M. S. (2020). Fluorescence lifetime patterns in macular telangiectasia type 2. Retina, 40(1), 99–108.
- Sallo, F. B., Leung, I., Chung, M., Wolf-Schnurrbusch, U. E. K., Dubra, A., Williams, D. R., Clemons, T., Pauleikhoff, D., Bird, A. C., & Peto, T. (2011). Retinal crystals in type 2 idiopathic macular telangiectasia. Ophthalmology, 118(12), 2461–2467.
- Tzaridis, S., Heeren, T., Mai, C., Thiele, S., Holz, F. G., Charbel Issa, P., & Herrmann, P. (2019). Right-angled vessels in Macular Telangiectasia Type 2. British Journal of Ophthalmology, 105(9), 1289–1296.
- Tzaridis, S., Hess, K., Heeren, T. F., Bonelli, R., Holz, F. G., & Friedlander, M. (2021). Hyperreflectivity on optical coherence tomography in Macular Telangiectasia Type 2. Retina, 41(7), 1428–1437.
- Zeimer, M., Gutfleisch, M., Heimes, B., Spital, G., Lommatzsch, A., & Pauleikhoff, D. (2015). Association between changes in macular vasculature in optical coherence tomography- and fluorescein- angiography and distribution of macular pigment in type 2 idiopathic macular Telangiectasia. Retina, 35(11), 2307–2316.
- Maesa, J., Baños‐Álvarez, E., Rosario‐Lozano, M., & Blasco‐Amaro, J. (2021). Diagnostic accuracy of optical coherence tomography angiography in the detection of neovasculature in age‐related Macular Degeneration: A meta‐analysis. Acta Ophthalmologica, 100(2).
- Wong, W. T., Forooghian, F., Majumdar, Z., Bonner, R. F., Cunningham, D., & Chew, E. Y. (2009). Fundus autofluorescence in type 2 idiopathic macular telangiectasia: Correlation with optical coherence tomography and microperimetry. American Journal of Ophthalmology, 148(4), 573–583.
- Pauleikhoff, L., Heeren, T. F. C., Gliem, M., Lim, E., Pauleikhoff, D., Holz, F. G., Clemons, T., Balaskas, K., Egan, C. A., & Charbel Issa, P. (2021). Fundus autofluorescence imaging in Macular Telangiectasia Type 2: MacTel study report number 9. American Journal of Ophthalmology, 228, 27–34.
- Breazzano, M. P., Yannuzzi, L. A., & Spaide, R. F. (2021). Genesis of retinal–Choroidal Anastomosis in macular telangiectasia type 2. Retina, 41(3), 464–470.
- Okada, M., Robson, A. G., Egan, C. A., Sallo, F. B., Esposti, S. D., Heeren, T. F., Fruttiger, M., & Holder, G. E. (2018). Electrophysiological Characterization of Macular Telangiectasia Type 2 and structure–function correlation. Retina, 38(1), 33–42.
- Wu, L. (2015). Multimodality Imaging in macular telangiectasia 2: A clue to its pathogenesis. Indian Journal of Ophthalmology, 63(5), 394–398.
- Powner, M. B., Gillies, M. C., Tretiach, M., Scott, A., Guymer, R. H., Hageman, G. S., & Fruttiger, M. (2010). Perifoveal Müller cell depletion in a case of Macular Telangiectasia Type 2. Ophthalmology, 117(12), 2407–2416.
- Chen, J. J., Sohn, E. H., Folk, J. C., Mahajan, V. B., Kay, C. N., Boldt, H. C., & Russell, S. R. (2014). Decreased macular thickness in nonproliferative macular telangiectasia type 2 with oral carbonic anhydrase inhibitors. Retina, 34(7), 1400–1406.
- Khodabande, A., Roohipoor, R., Zamani, J., Mirghorbani, M., Zolfaghari, H., Karami, S., & Modjtahedi, B. S. (2019). Management of Idiopathic Macular Telangiectasia Type 2. Ophthalmology and Therapy, 8(2), 155–175.
- Pasquin, S., Sharma, M., & Gauchat, J.-F. (2015). Ciliary neurotrophic factor (CNTF): New facets of an old molecule for treating neurodegenerative and metabolic syndrome pathologies. Cytokine & Growth Factor Reviews, 26(5), 507–515.
- Yong, J., Groeger, S., von Bremen, J., & Ruf, S. (2022). Ciliary neurotrophic factor (CNTF) and its receptors signal regulate cementoblasts apoptosis through a mechanism of ERK1/2 and Caspases Signaling. International Journal of Molecular Sciences, 23(15), 8335.
- Chew, E. Y., Clemons, T. E., Jaffe, G. J., Johnson, C. A., Farsiu, S., Lad, E. M., Guymer, R., Rosenfeld, P., Hubschman, J.-P., Constable, I., Wiley, H., Singerman, L. J., Gillies, M., Comer, G., Blodi, B., Eliott, D., Yan, J., Bird, A., & Friedlander, M. (2019). Effect of ciliary neurotrophic factor on retinal neurodegeneration in patients with Macular Telangiectasia Type 2. Ophthalmology, 126(4), 540–549.