GLP-1 Receptor Agonists in Neuro-Ophthalmic Disease

Welcome to the “Neuro Nuggets” column within the Journal of Medical Optometry (JoMO)! This column aims to make neuro-ophthalmic disease more approachable by blending real-world clinical cases with evidence-based medicine. The patients in this edition’s column demonstrate the relevance of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) in the realm of neuro-ophthalmic disease. Enjoy!
Case #1
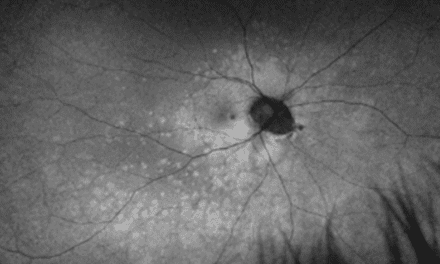
A 44-year-old male with a history of obesity (BMI 42.8), hypertension, migraine headaches for over 20 years, and obstructive sleep apnea presented to the eye clinic to establish care. The patient endorsed a vague, mild visual blur of chronic duration with no acute worsening. He had a chronic headache history but denied any recent exacerbations. He denied tinnitus and nausea/vomiting. On exam, best-corrected visual acuity was 20/20 in each eye. Color vision was intact bilaterally. Pupils were equal, round, and reactive to light without a relative afferent pupillary defect. Contrast sensitivity (MARS) was intact at 1.64 logCS in each eye. Extraocular motility and alignment testing was within normal limits. Automated perimetry showed mildly enlarged blind spots but was otherwise intact in all quadrants of each eye. Slit lamp examination was unremarkable, and intraocular pressures were normotensive and symmetric. On dilated fundus examination, there was evidence of bilateral optic disc edema with the right eye worse than the left eye; choroidal folds were evident in the right eye posterior pole (Figure 1). The retinal vasculature was mildly tortuous but otherwise unremarkable, and there were no retinal or choroidal abnormalities.
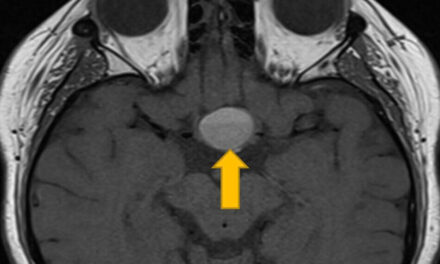
Same-day MRI brain and orbits with MRV (magnetic resonance venography) was ordered to rule out any space-occupying lesions or cerebral venous sinus thrombosis (CVST). MRI/MRV showed partially empty sella turcica and mild bilateral posterior flattening of the globes (Figure 2) with focal stenoses at the transverse and sigmoid sinuses, but there were no mass lesions or CVST noted. The patient also underwent various blood tests, all of which were unremarkable. Lumbar puncture (LP) demonstrated an opening pressure of 32.5 cm H2O, and the subsequent cerebrospinal fluid (CSF) analysis was unremarkable. The constellation of papilledema, normal MRI/MRV, elevated opening pressure by LP, and normal CSF analysis enabled the diagnosis of idiopathic intracranial hypertension (IIH).
The patient was started on oral acetazolamide 250 mg twice per day. The clinical pharmacy team was consulted with consideration of initiating semaglutide, in addition to diet and exercise strategies, to optimize the patient’s weight. The patient was ultimately started on semaglutide subcutaneous injections 0.25 mg weekly.
At follow-up three months after initial presentation, the patient already had approximately 15 pounds of weight loss. The bilateral optic disc edema was notably improved in each eye (Figures 3 and 4), and the visual parameters remained intact in each eye.
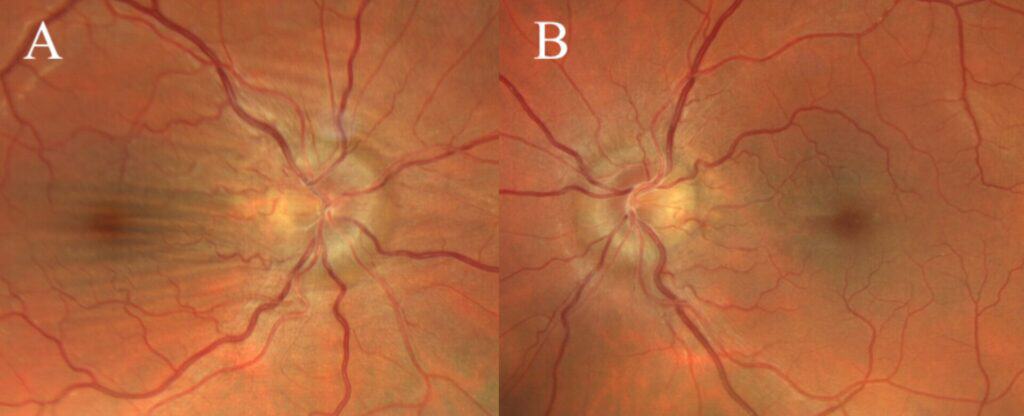
Figures 1A&B. Baseline fundus photos demonstrating optic disc edema in the right eye (A) and the left eye (B) with choroidal folds in the right eye.
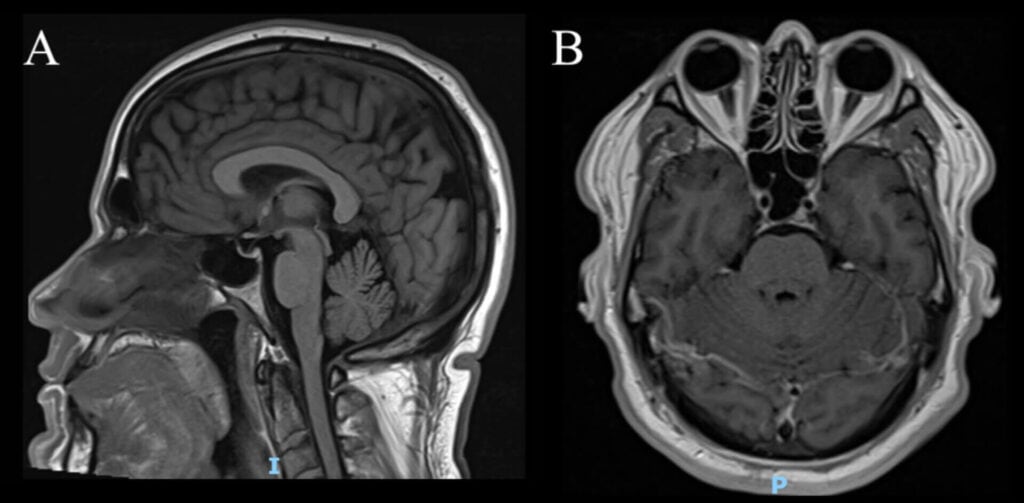
Figure 2. (A) Sagittal view of brain MRI demonstrating partially empty sella and (B) Axial view of brain MRI showing bilateral mild posterior globe flattening.
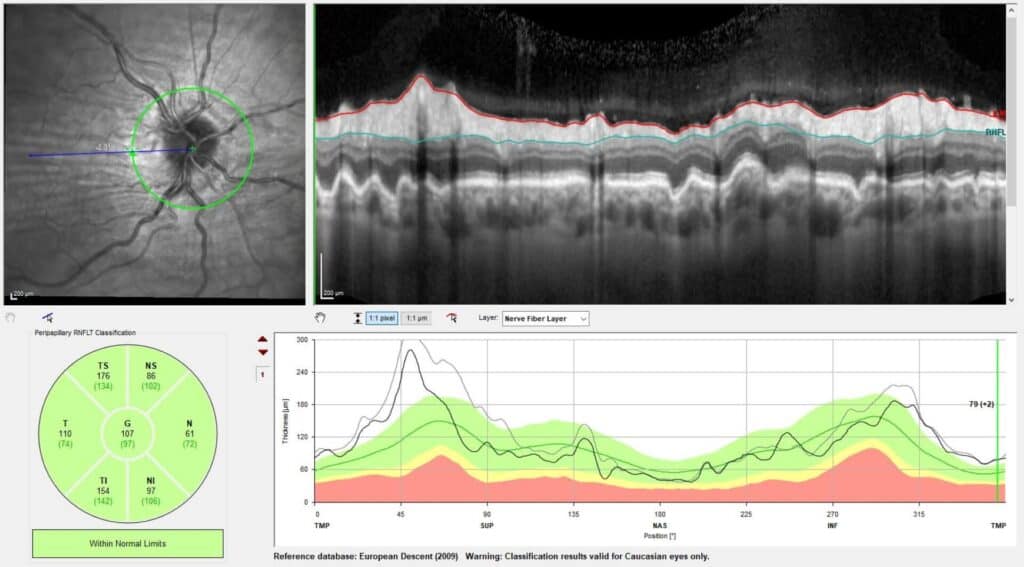
Figure 3. Peripapillary retinal nerve fiber layer (pRNFL) optical coherence tomography (OCT) imaging demonstrating decreased pRNFL thickness from baseline (black line) to six-week follow-up (gray line) for the right eye.
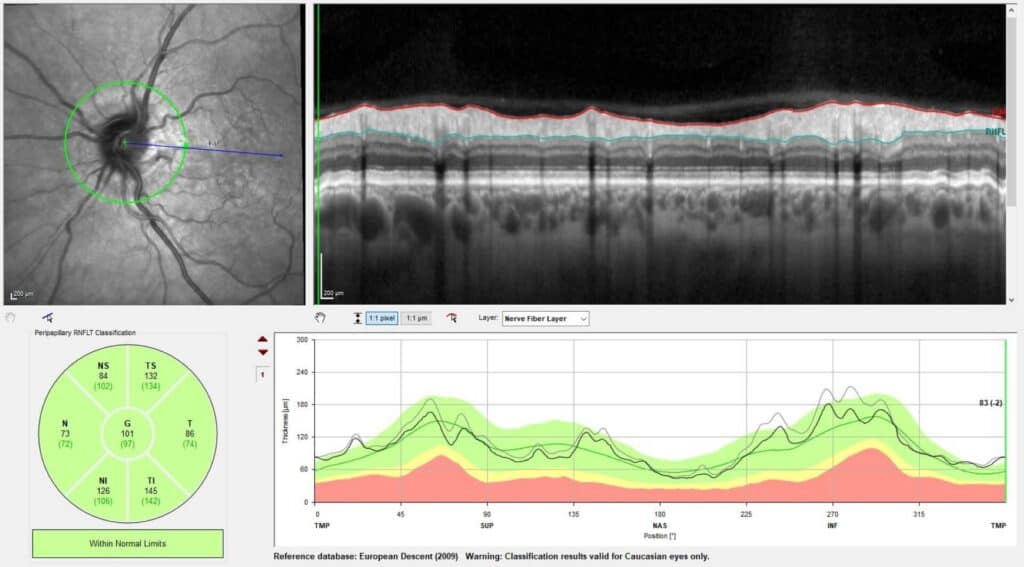
Figure 4. Peripapillary retinal nerve fiber layer (pRNFL) optical coherence tomography (OCT) imaging demonstrating decreased pRNFL thickness from baseline (black line) to six-week follow-up (gray line) for the left eye.
Case #2
A 54-year-old male with a history of hypertension, pre-diabetes, obstructive sleep apnea, erectile dysfunction, and obesity (BMI 37.8) presented with no acute visual concerns. The patient was managed on amlodipine, lisinopril, sildenafil, and semaglutide subcutaneous injections, recently uptitrated from 1.7 mg to 2.4 mg weekly. The patient reported taking blood pressure medications in the morning and admitted to sub-optimal adherence with his recommended continuous positive airway pressure (CPAP) device.
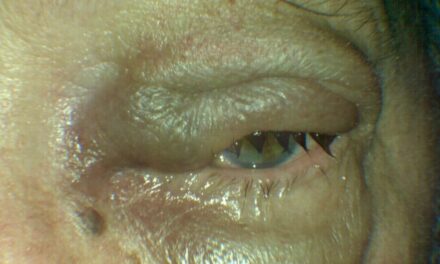
On exam, best-corrected visual acuity was 20/20 in each eye. Pupils, color vision, and contrast sensitivity testing were all normal or unremarkable. Extraocular motility and alignment testing was within normal limits, as were slit lamp examination and intraocular pressure. Dilated fundus evaluation demonstrated notable optic disc edema, mostly concentrated superiorly/superonasally in the right eye (Figure 5); optic nerve evaluation was normal in the left eye, aside from small/crowded optic nerve head anatomy. Automated perimetry (HVF 24-2 SITA Fast) showed a mild inferior nasal defect in the right eye, and an intact visual field in the left eye. The patient denied any signs/symptoms that would be concerning for giant cell arteritis and denied eye pain that might suggest optic neuritis or an alternate etiology. The clinical presentation was determined to be consistent with non-arteritic anterior ischemic optic neuropathy (NA-AION) in the right eye.
No treatment was recommended, but the patient was counselled on the potential risk of his medications contributing to his presentation. After consultation with clinical pharmacy providers, the patient elected to discontinue both semaglutide and sildenafil. The patient was also encouraged to use CPAP more regularly. At follow-up six weeks and three months after presentation, the patient’s exams demonstrated gradual improvement in right optic disc edema while maintaining intact visual acuity, visual fields, and ganglion cell layer integrity by optical coherence tomography (OCT) (Figure 6).
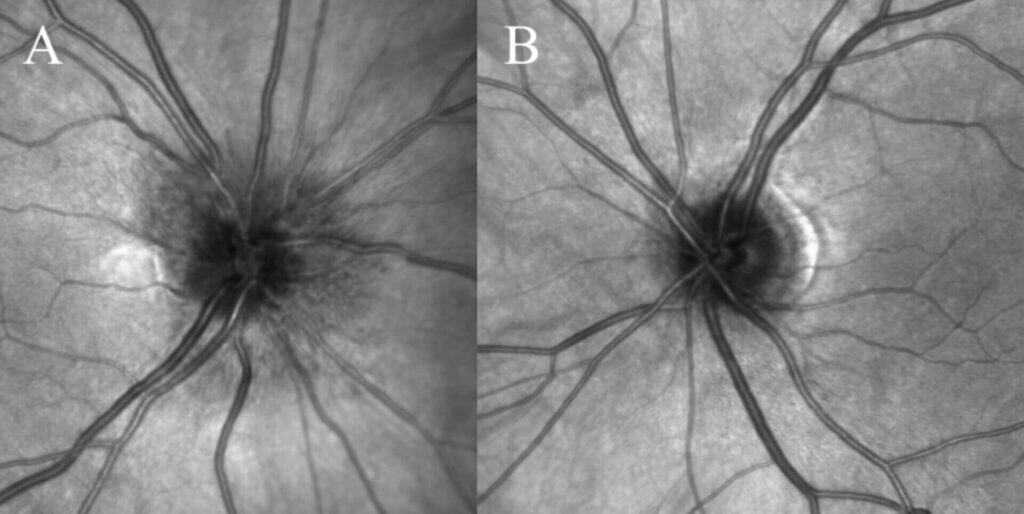
Figures 5A&B. Infrared imaging demonstrating peripapillary retinal nerve fiber layer edema right eye (A) and disc-at-risk optic nerve anatomy without edema left eye (B).
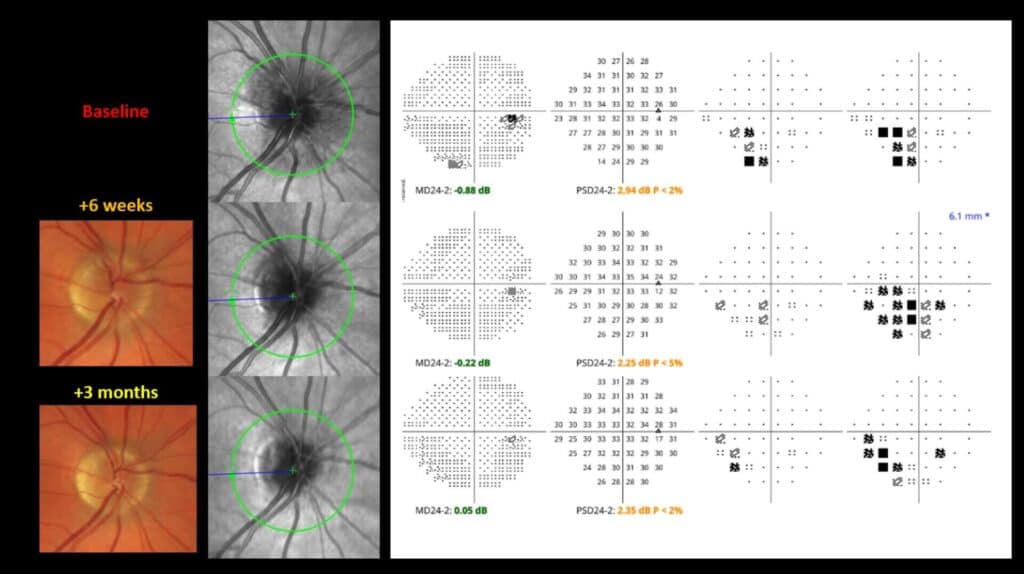
Figure 6. Serial testing for right eye NA-AION demonstrating gradual improvement in optic disc edema with similar visual field testing across different time points.
Discussion
Case #1 demonstrates the potential role that GLP-1 RAs can play in helping IIH patients optimize their weight and, therefore, mitigate their risk of vision loss. Glucagon-like peptide 1 is a hormone produced by intestinal cells and also neurons in the brain.1 GLP-1 RAs have demonstrated a benefit in patients with diabetes by stimulating glucose-induced insulin release, inhibiting the secretion of glucagon, and suppressing appetite.1 Since body weight is one of the main modifiable risk factors for IIH, this clearly has relevance to eye care providers. Multiple studies have indicated the benefit of these medications in patients with IIH.2-4 One study even showed a benefit of these medications beyond just the resultant weight loss. A study by Mitchell et al used a surgically implanted intracranial pressure (ICP) monitor and showed reduction in ICP within minutes to hours after GLP-1 RA initiation – long before weight loss would have occurred.1 GLP-1 receptors are expressed in the human choroid plexus, and it is believed that GLP-1 RAs reduce CSF secretion which leads to lower ICP.1,5 Importantly, this patient’s weight loss was likely multifactorial (diet, exercise, semaglutide) as was the improvement in bilateral optic disc edema (weight loss, acetazolamide, semaglutide). Though data is still emerging, it appears that GLP-1 RAs show promise as a potential adjunct medication for IIH patients with benefits that extend beyond weight loss.
Case #2 highlights the role of recognizing potential risk factors for NA-AION. Though the precise pathophysiology is still the subject of debate, NA-AION has been described as a circulatory or microvascular disorder for which a compartment syndrome (“disc-at-risk”) may play a role.6 Multiple medications have been implicated as contributing to risk of NA-AION, including amiodarone and phosphodiesterase inhibitors, though the link is controversial.7 Recently, a retrospective review of over 16,000 patients presenting to a tertiary neuro-ophthalmology practice showed a potential link between semaglutide and NA-AION.8 However, other studies have not demonstrated a similar link,9 and the overall risk remains low (around one case of NA-AION in every several thousand individuals prescribed GLP-1 RAs), and the variability in design across studies makes definitive conclusions challenging. The precise connection linking GLP-1 RAs to NA-AION is unclear though some have postulated volume depletion, hypotension, transient microvascular dysregulation, and impaired vascular autoregulation as potential mechanisms.10
Since the patient in Case #2 was asymptomatic and the optic disc edema was an incidental finding, it is difficult to know exactly how long the optic disc edema was present prior to presentation. There are cases of mild or “incipient” NA-AION in the ophthalmic literature whereby the optic nerve remains swollen for a period of time before any infarction or symptomatic visual loss occurs.11 This presents an opportunity for eye care providers. Early recognition and intervention (in this case, discontinuation of potentially contributing medications) may help to guard against worsening visual loss in cases of mild or incipient NA-AION.11
Clinical Pearls
- Patients should be counselled on the potential effects of their systemic medications on their ocular health and vision
- GLP-1 receptor agonists may serve an adjunct role in helping patients with IIH achieve optimal weight and guard against visual loss
- The current ophthalmic literature is varied with regard to GLP-1 receptor agonists and risk of NA-AION, and clinicians must stay informed as data continues to emerge on this topic
References
- Mitchell JL, Lyons HS, Walker JK, et al. The effect of GLP-1RA exenatide on idiopathic intracranial hypertension: a randomized clinical trial. Brain: A J Neurol. 2023;146(5):1821-1830.
- Sioutas GS, Mualem W, Reavey-Cantwell J, Rivet DJ 2nd. GLP-1 Receptor Agonists in Idiopathic Intracranial Hypertension. JAMA Neurol. 2025 Sep 1;82(9):887-894. doi: 10.1001/jamaneurol.2025.2020. PMID: 40658395; PMCID: PMC12261113.
- Ognard J, Alipour Khabir S, Ghozy S, El Hajj G, Kallmes KM, Chen JJ, Kadirvel R, Kallmes DF, Brinjikji W. Use of glucagon-like peptide-1 receptor agonists in idiopathic intracranial hypertension : a systematic review. J Headache Pain. 2025 Oct 7;26(1):202. doi: 10.1186/s10194-025-02148-3. PMID: 41057780; PMCID: PMC12502165.
- Ahmad J, Hamdy AM, Elfakharany B, Elsharkawy MM, El-Samahy M, Shehata MM, Moubarak ES. The effect of GLP-1 agonist on idiopathic intracranial hypertension: a systematic review and meta-analysis. Ther Adv Neurol Disord. 2025 Oct 27;18:17562864251378845. doi: 10.1177/17562864251378845. PMID: 41180125; PMCID: PMC12575947.
- Krajnc N et al. Treatment with GLP-1 receptor agonists is associated with significant weight loss and favorable headache outcomes in idiopathic intracranial hypertension. J Headache Pain. 2023 Jul 18;24(1):89. doi: 10.1186/s10194-023-01631-z. PMID: 37460968; PMCID: PMC10353241.
- Rizzo JF 3rd. Unraveling the Enigma of Nonarteritic Anterior Ischemic Optic Neuropathy. J Neuroophthalmol. 2019 Dec;39(4):529-544. doi: 10.1097/WNO.0000000000000870. PMID: 31790002.
- Kaur K, Margolin E. Nonarteritic Anterior Ischemic Optic Neuropathy. [Updated 2025 Sep 14]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK559045/
- Hathaway JT, Shah MP, Hathaway DB, Zekavat SM, Krasniqi D, Gittinger JW Jr, Cestari D, Mallery R, Abbasi B, Bouffard M, Chwalisz BK, Estrela T, Rizzo JF 3rd. Risk of Nonarteritic Anterior Ischemic Optic Neuropathy in Patients Prescribed Semaglutide. JAMA Ophthalmol. 2024 Aug 1;142(8):732-739. doi: 10.1001/jamaophthalmol.2024.2296. PMID: 38958939; PMCID: PMC11223051.
- Abbass et al. The Effect of Semaglutide and GLP-1 RAs on Risk of Nonarteritic Anterior Ischemic Optic Neuropathy. Am J Ophthalmol. 2025 Jun;274:24-31. doi: 10.1016/j.ajo.2025.02.025. Epub 2025 Feb 25. PMID: 40015592.
- Heberer K, Bress AP, Cogill S, Maldonado AI, Kim SH, Nallamshetty S, Chen YQ, Shih MC, Lynch JA, Lee JS. New-Onset Nonarteritic Anterior Ischemic Optic Neuropathy and Initiators of Semaglutide in US Veterans With Type 2 Diabetes. JAMA Ophthalmol. 2026 Feb 12:e256262. doi: 10.1001/jamaophthalmol.2025.6262. Epub ahead of print. PMID: 41678180; PMCID: PMC12902922.
- Lam WZ, Shen M, Zhang Q, Le VH, Singla K, Fooladi MI, Jiang H, Lam BL, Wang RK, Gregori G, Rosenfeld PJ. Longitudinal widefield OCT optic nerve thickness measurements in a case of incipient non-arteritic ischemic optic neuropathy. Am J Ophthalmol Case Rep. 2025 Jun 27;39:102367. doi: 10.1016/j.ajoc.2025.102367. PMID: 40686763; PMCID: PMC12271809.
Dr. Kane graduated from New England College of Optometry in 2015 and went on to complete an ocular disease/primary care residency at VA Boston Jamaica Plain from 2015-2016. He is currently an attending optometrist at VA Boston. His interests include clinical teaching, neuro-ophthalmic disease, retinal vascular disease, glaucoma, and ocular manifestations of systemic disease.