An optometrically driven approach: diagnosing a patient with active thyroid eye disease in the setting of Hashimoto’s thyroiditis

Abstract
Background: TED can mimic a variety of other conditions, but a directed clinical exam combined with laboratory and radiologic studies allows the clinician to arrive at the correct diagnosis and coordinate timely intervention.
Case Report: 57-year-old white female presented as a new patient for an eye exam with intermittent binocular diplopia, extraocular motility restrictions, and bilateral proptosis. Clinical exam combined with laboratory testing and magnetic resonance imaging (MRI) of brain/orbits aided in arriving at the diagnosis of active thyroid eye disease (TED). Interdisciplinary collaboration facilitated treatment with recently FDA-approved teprotumumab infusions. Teprotumumab can be beneficial for TED patients, but not without potential side effects, including the possibility of permanent hearing loss.
Conclusion: This case report highlights optometry’s role in diagnosing and facilitating appropriate management for TED patients to prevent potentially devastating vision loss.
Keywords: thyroid eye disease, Hashimoto’s thyroiditis, diplopia, proptosis, teprotumumab
Introduction
Thyroid eye disease (TED), also known as thyroid-associated ophthalmopathy (TAO), is a debilitating autoimmune condition that causes damage to different structures within the orbit. It is a disease that predominantly occurs in women with thyroid dysfunction.1 Thyroid eye disease may precede or follow endocrine manifestations; when present, it typically occurs within 18 months of a thyroid diagnosis.2 The disease course can range from mild to severe and is challenging to diagnose. When not treated early on, it can be a life-altering disease that leaves patients with permanent vision loss, disfigurement, and eye muscle restrictions. TED most often manifests in patients with Graves’ disease or hyperthyroidism. Uncommonly, TED can occur in patients with hypothyroidism. Hashimoto’s thyroiditis is the most common cause of hypothyroidism in adults who are iodine sufficient.3 This is a case report of a patient who initially presented to the eye clinic and was ultimately diagnosed with TED secondary to previously undiagnosed Hashimoto’s thyroiditis.
Case Report
A 57-year-old white female presented for an initial eye exam complaining of intermittent episodes of binocular horizontal diplopia (ongoing for the past 2-3 months), worse at distance than near (with or without correction) and often noticed first thing in the morning upon waking. During these episodes, she associated a dull ache behind her left eye, described as a headache. She denied constant diplopia, and there was no recent history of head trauma. She also denied any other associated neurological symptoms. Her medical history was significant for obstructive sleep apnea and hypothyroidism, diagnosed two years earlier. There was no history of thyroid surgery or radioactive iodine therapy (RAI). She denied a history of hyperthyroidism or Grave’s disease. For medications, she was taking albuterol, loratadine, and levothyroxine. The patient had recently been working with her primary care provider to adjust levothyroxine to the appropriate dose, and by the time we saw her in the eye clinic she was euthyroid. Also of note, she was a former smoker with greater than 30 pack years and quit smoking two years prior.
Upon examination, the patient’s best-corrected Snellen distance visual acuity was 20/25 OD and 20/20 OS. Pupils were equal (in dim and bright light), round, and reactive to light without a relative afferent pupillary defect. Formal Humphrey visual field testing was reliable and clear without visual field defects in either eye. There was no dyschromatopsia. Cover test showed an intermittent right hypertropia that was non-comitant by Parks 3-step testing but did not map to a specific extraocular muscle that would point to a cranial nerve palsy. Extraocular motility (EOM) testing revealed mild discomfort on eye movements with 10% right and left abduction deficits, ~10% down-gaze limitation of the right eye, ~20% up-gaze limitation of the left eye, and right inferior oblique over-action (Figure 1a). Externally, the patient had a noticeable left head tilt and left upper eyelid retraction. The patient reported diplopia in all positions of gaze aside from primary gaze. Slightly asymmetric proptosis was documented by Hertel exophthalmometry (base 95mm; OD 21mm, OS 20mm). Anterior segment exam was significant for a small, resolving subconjunctival hemorrhage of the right eye, and plica injection was noted in both eyes, but was otherwise normal. On dilated exam, there was a resolving flame-shaped hemorrhage within 1DD superior nasal to the right optic nerve head. Both optic nerves were well-perfused with healthy pink rim tissue and showed no signs of optic nerve compression. In summary, the patient exhibited multiple findings suggestive of thyroid eye disease, which is somewhat atypical in the setting of hypothyroidism without a history of RAI or Graves’ disease.
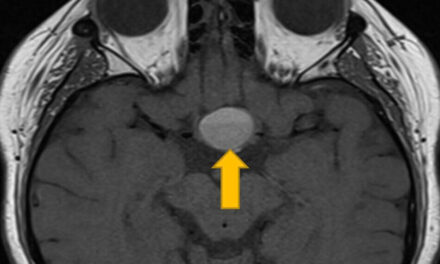
Despite hypothyroidism not being the most common cause of TED, the clinical picture is convincing: a middle-aged female patient, with diplopia worse in the morning consistent with EOM abnormalities that did not map to a specific pattern, dull pain with eye movement, eyelid retraction, and mild proptosis. Other differentials at this point also included a decompensating congenital right cranial nerve (CN) 4 palsy with spread of comitance, decompensating right hyperphoria, myasthenia gravis (due to reported variability/fluctuation and EOM abnormalities), an unlikely but possibly an evolving ischemic CN 3,4 or 6 palsy. Given the full clinical picture, we were strongly suspicious for TED. As the next step of the work-up, myasthenia labs and an MRI of the patient’s brain/orbits were ordered to help further classify the EOM abnormalities. Myasthenia labs returned normal. MRI of the brain and orbits demonstrated diffuse extraocular muscle enlargement with T2 hyperintensity and enhancement involving the right greater than left orbits, with notable sparing of the tendinous insertions (Figure 2). Differentials based on the MRI results included thyroid eye disease (most likely), idiopathic orbital inflammation, IgG4 disease, sarcoidosis, and lymphoma (unlikely).
Endocrinology was consulted for further characterization of her thyroid dysfunction. Of note, she had a thyroid ultrasound with her primary care provider earlier in the year that showed possible thyroiditis. Endocrinology service evaluated the patient and ordered labs which were significant for thyroglobulin antibodies (Tgab), thyroid stimulating IgGs (TSI), thyroid peroxidase (TPO) antibodies, and thyroid stimulating hormone receptor (TSHR) antibodies (TRAb). Thyroid total T3 and TSH were normal. Endocrinology determined that the patient likely had a competing process of thyroid overactivity and thyroid destruction from the TPO antibodies, with the destructive antibodies “winning out” and leading to hypothyroidism. Given that she has multiple antibodies, even though she was clinically and biochemically euthyroid, she was found to have significant thyroid autoimmunity, which is suggestive of Hashimoto’s thyroiditis. Next, we connected the patient with the Oculoplastics team for consideration of Teprotumumab infusions to treat her active TED and proptosis. We also helped coordinate an audiology evaluation prior to Teprotumumab infusions due to the potential side effect of hearing loss; her baseline hearing was normal.
Discussion
TED is a multifactorial autoimmune disease with an incidence of 16 per 100,000 in females and 2.9 per 100,000 in males.9 Risk factors include female gender, middle age, smoking, and other autoimmune diseases.9 The pathophysiology of TED is not entirely understood, but is more recently thought to be due to CD34+ fibrocytes that infiltrate the orbit and express high levels of TSH receptors.12 The primary autoantigen involved in TED is TSHR antibodies because they are present on orbital fibroblasts and adipocytes. TSHR antibodies activate orbital fibroblasts TSHRs, adipocyte TSHRs, and insulin-like growth factor 1 receptor (IGF-1) receptors, which stimulate an orbital inflammatory environment. Hydrophilic glycosaminoglycans (GAG) accumulate, leading to muscle and tissue swelling in the orbit. This results in extraocular muscle dysfunction and enlargement, proptosis, and periorbital swelling.
TED in the setting of Hashimoto’s thyroiditis is rare (up to 6%)5, and the risk increases with a longer duration of Hashimoto’s disease. The incidence of Hashimoto’s has increased in the last three decades with an incidence rate of 0.15-0.3 cases per 1000.6 Hashimoto’s thyroiditis, also known as chronic autoimmune thyroiditis, is characterized by gradual thyroid failure due to lymphocytic infiltration and autoimmune facilitated destruction of the thyroid.7 The exact etiology of Hashimoto’s is not entirely understood either; however, it is thought to result from a combination of genetic elements, environmental factors, and epigenetic influences.6 There are many antibodies involved in autoimmune thyroiditis. A majority of those with Hashimoto’s thyroiditis have high serum levels of antibodies to thyroglobulin (Tg) and thyroid peroxidase (TPO).
Diagnosing Hashimoto’s begins with confirming hypothyroidism with serum thyroid function tests. High serum TSH levels and low free T4 are diagnostic for hypothyroidism. Once a patient is confirmed to be hypothyroid, more specific antithyroid antibodies, such as, thyroglobulin and thyroid peroxidase antibodies are screened for. The patient in this case report had positive TPO antibodies, TSI antibodies, TRAb, and Tgabs. The thinking is that she likely had competing processes of thyroid overactivity and thyroid destruction from the TPO antibodies, with the destructive antibodies “winning out” and leading to hypothyroidism.
Although TED is seen in about 40-50% of patients with Graves’ disease, it is important to also consider TED in patients with hypothyroidism.8,10 Signs of TED may precede symptoms and can include upper eyelid retraction, lid lag, conjunctival and caruncle injection, eyelid edema, extraocular muscle dysfunction and proptosis. Symptoms are typically non-specific and related to dry eye, such as foreign body sensation or tearing. However, they can also include retrobulbar discomfort, diplopia and, in the worst case, vision loss from optic nerve compression.2 In this case report, the patient displayed left upper eyelid retraction and caruncle injection. However, the more prominent finding that ultimately led to the presumed diagnosis of active thyroid eye disease was the EOM dysfunction that was worse in the morning and did not follow a specific pattern for other cranial nerve palsies. In addition to the clinical exam, lab work and orbital imaging are required for diagnosis.
Cross-sectional imaging for TED can be performed with either contrast-enhanced computerized tomography (CT) or MRI. Typical imaging findings include enlargement and hyperenhancement of the extraocular muscles, as well as increased intramuscular fat, with sparing of the tendinous insertions.14 Increased retro-ocular fat may also be present.14 Bilateral involvement of these CT findings is common (greater than 75%).14 In decreasing order of frequency, TED typically affects the levator palpebrae superioris, inferior rectus, medial rectus, superior rectus, lateral rectus, and lastly the superior/inferior oblique muscles.15,16
It is important to determine the stage of disease activity and severity of TED to guide management. There are multiple grading systems for TED, including NOSPECS classification (No signs or symptoms, Only signs, Soft tissue involvement, Proptosis, Extraocular muscle involvement, Corneal involvement, and Sight loss due to optic nerve compression) and the Clinical Activity Score (CAS), which is used in this case. The CAS is based on a list of inflammatory signs and symptoms, for which each parameter is worth one point. The disease is considered clinically active if the total score is four or greater. Initially, this patient’s CAS was four due to the presence of spontaneous orbital pain, gaze-evoked orbital pain, eyelid erythema and inflammation of the caruncle or plica.
Treatment and Management
Optimal management of TED is determined by a multidisciplinary team of eye care providers, primary care, and endocrinology. The first part of treatment includes the management of systemic thyroid disease and smoking cessation, if applicable. Smoking increases the risk of TED by 2.47x and is the most important modifiable risk factor in the prevention and progression of TED.2 Thyroid medications do not typically improve TED.9 Management of TED is based on whether the disease is active or inactive and the level of disease severity. Traditionally, the first line of treatment has been systemic corticosteroids. However, given the side effects of oral steroids, other biologic agents are sometimes more readily used. Unless compressive optic neuropathy is present, surgery is only pursued once the disease is inactive.
Teprotumumab is a new treatment option for patients with active thyroid eye disease. It is a monoclonal antibody inhibitor of insulin-like growth factor 1 receptor (IGF-IR). 10 The strong link and crosstalk between TSHR and the IGF-1 receptor forms the basis for teprotumumab and plays a critical role in reducing the extraocular muscle and orbital fat volumes, leading to a reduction in diplopia and proptosis.10 Teprotumumab is administered as an intravenous infusion given once over three weeks for a total of 8 infusions.10 Improvement in proptosis response, CAS and diplopia response are seen as early as six weeks and can continue throughout the 24-week course.10 Safety and efficacy were studied in phase II and III clinical trials on patients with active, moderate-to-severe TED. The phase II study showed improvement of proptosis by ≥2mm in 79% of treated patients, compared to 22% of placebo patients.13 Also, 69% of patients who received teprotumumab had a clinical activity score of 0 or 1 at week 24, compared with 21% who received a placebo.13 Adverse events include anaphylaxis, infusion-related reactions, hyperglycemia, muscle spasms, diarrhea and potentially irreversible hearing impairment10 In the phase III trial, five patients in the teprotumumab group were documented to have various forms of hearing impairment, fortunately all of which resolved without permanent hearing dysfunction.10 IGF-1 and IGF-1R are important in maintaining otologic health, likely accounting for potential hearing dysfunction secondary to teprotumumab treatment.17 A small case review of 28 patients treated with teprotumumab identified four patients who developed hearing dysfunction, two resulting in permanent hearing damage.17 Given the risk for potentially permanent audiologic dysfunction, educating patients and considering formal baseline auditory testing pre-, during, and post-treatment with teprotumumab is important.17 Collaborating with audiology during teprotumumab infusions should be considered, along with encouraging patients to report any new symptoms of potential audiologic dysfunction, including tinnitus, autophony, hypoacusis, hyperacusis, or fullness of the ear.17 Teprotumumab can substantially improve symptoms for TED patients, but is not without potential side effects including possible permanent hearing loss.
Currently, this patient has completed seven out of eight infusions of teprotumumab. Her CAS is now zero and she is no longer experiencing diplopia or periorbital pain. Repeated formal visual fields have also shown no defects consistent with optic neuropathy. Identification of active TED with subsequent initiation of Teprotumumab and maintenance of levothyroxine resulted in a significant improvement of the clinical disease state, symptom improvement, and overall better quality of life.
Conclusion
Thyroid Eye Disease can be a challenging diagnosis. Still, a directed clinical exam combined with laboratory and radiologic studies allows a clinician to arrive at the correct diagnosis and coordinate timely and appropriate intervention. Primary eye care providers are on the front line when patients present with eye findings suspicious for TED. Although rare, TED due to Hashimoto’s thyroiditis should be considered. Multidisciplinary collaboration is required to manage patients with TED. Teprotumumab can offer TED patients a significant improvement in quality of life and a reduction in disease severity.
References
- Davies, TF., Burch, HB. Clinical features and diagnosis of thyroid eye disease. In:UpToDate, Post, TW (Ed), UpToDate, Boston,MA, 2023.
- Weiler DL. Thyroid eye disease: a review. Clin Exp Optom. 2017 Jan;100(1):20-25. doi: 10.1111/cxo.12472. Epub 2016 Oct 4. PMID: 27701774.
- Ragusa F., Fallahi P., et al. Hashimotos’ thyroiditis: Epidemiology, pathogenesis, clinic and therapy. Best Practive & Research Clinical Endo. 2019;33(6). Doi: 10.1016/j.beem.2019.101367.
- Bartley GB. The epidemiologic characteristics and clinical course of ophthalmopathy associated with autoimmune thyroid disease in Olmsted County, Minnesota. Trans Am Ophthalmol Soc. 1994;92:477-588. PMID: 7886878; PMCID: PMC1298522.
- Kahaly GJ, et al. Thyroid Stimulating Antibodies Are Highly Prevalent in Hashimoto’s Thyroiditis and Associated Orbitopathy. J Clin Endocrinol Metab. 2016 May;101(5):1998-2004. doi: 10.1210/jc.2016-1220. Epub 2016 Mar 10.
- Ralli M, Angeletti D, Fiore M, D’Aguanno V, Lambiase A, Artico M, de Vincentiis M, Greco A. Hashimoto’s thyroiditis: An update on pathogenic mechanisms, diagnostic protocols, therapeutic strategies, and potential malignant transformation. Autoimmun Rev. 2020 Oct;19(10):102649. doi: 10.1016/j.autrev.2020.102649. Epub 2020 Aug 15. PMID: 32805423.
- Mincer DL, Jialal I. Hashimoto Thyroiditis. [Updated 2022 Jun 21]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459262/
- Menconi, F., Marcocci, C., & Marinò, M. (2014). Diagnosis and classification of Graves’ disease. Autoimmunity reviews, 13(4-5), 398-402.
- Hennein L, Robbins SL. Thyroid-Associated Orbitopathy: Management and Treatment. J Binocul Vis Ocul Motil. 2022 Jan-Mar;72(1):32-46. Epub 2021 Dec 7. PMID: 34874796.
- Douglas RS, et al. Teprotumumab for the Treatment of Active Thyroid Eye Disease. N Engl J Med. 2020 Jan 23;382(4):341-352. doi: 10.1056/NEJMoa1910434.
- Kossler A, Sears CM, Dosiou C. Hearing Loss and Teprotumumab. J Endocr Soc. 2021 May 3;5(Suppl 1):A839. doi: 10.1210/jendso/bvab048.1713. PMCID: PMC8089925.
- Men CJ, Kossler AL, Wester ST. Updates on the understanding and management of thyroid eye disease. Ther Adv Ophthalmol. 2021 Jun 30;13:25158414211027760. doi: 10.1177/25158414211027760. PMID: 34263138; PMCID: PMC8252358.
- Smith TJ, Kahaly GJ, Ezra DG, Fleming JC, Dailey RA, Tang RA, Harris GJ, Antonelli A, Salvi M, Goldberg RA, Gigantelli JW, Couch SM, Shriver EM, Hayek BR, Hink EM, Woodward RM, Gabriel K, Magni G, Douglas RS. Teprotumumab for Thyroid-Associated Ophthalmopathy. N Engl J Med. 2017 May 4;376(18):1748-1761. doi: 10.1056/NEJMoa1614949. PMID: 28467880; PMCID: PMC5718164.
- Parmar H, Ibrahim M. Extrathyroidal Manifestations of Thyroid Disease: Thyroid Ophthalmopathy. Neuroimaging Clin N Am. 2008 Aug;18(3):527-36.
- Nugent R, Belkin R, Neigel J et al. Graves Orbitopathy: Correlation of CT and Clinical Findings. Radiology. 1990;177(3):675-82.
- Davies M & Dolman P. Levator Muscle Enlargement in Thyroid Eye Disease-Related Upper Eyelid Retraction. Ophthalmic Plast Reconstr Surg. 2017;33(1):35-9.
- Belinsky, Irina M.D. Creighton, Francis X. Jr M.D. Mahoney, Nicholas M.D. Petris, Carisa K. M.D. Callahan, Alison B. M.D. Campbell, Ashley A. M.D. Kazim, Michael M.D. Lee, H. B. Harold M.D. Yoon, Michael K. M.D. Dagi Glass, Lora R. M.D. Teprotumumab and Hearing Loss: Case Series and Proposal for Audiologic Monitoring. Ophthalmic Plastic and Reconstructive Surgery 38(1):p 73-78, January/February 2022. | DOI: 10.1097/IOP.0000000000001995