Management of a Retinal Arterial Macroaneurysm

ABSTRACT
BACKGROUND
Retinal arterial macroaneurysms (RAMs) are acquired, focal dilations of a retinal artery, most often found in hypertensive patients. While patients with RAMs are often asymptomatic, providers must be able to clinically differentiate them from other retinal conditions so they can manage and counsel patients appropriately. This case report reviews the anatomical features, risk factors, clinical course, pathophysiology, and treatment options for potential sequelae of a RAM.
CASE REPORT
An 83-year-old white male with uncontrolled hypertension presented for an eye examination noting a mild and gradual increase in blur at distance and near in each eye with his glasses over the last two years. A hemorrhagic RAM was noted in the inferotemporal arcade of the right eye. The patient subsequently developed a vitreous hemorrhage and a circinate ring of exudates from the macroaneurysm. Co-management with the patient’s primary care provider yielded improved blood pressure control and resolution of the RAM and its sequelae.
CONCLUSION
While most RAMs have a benign course with spontaneous resolution, associated retinal changes may cause a reduction in vision and warrant treatment via direct or indirect photocoagulation, intravitreal injections of anti-vascular endothelial growth factor, pars plana vitrectomy, laser membranotomy, pneumatic displacement, or submacular surgery. Ultimately, patients with a RAM should also be evaluated for hypertension by a primary care physician as a strong correlation exists between the two conditions.
Keywords: retinal arterial macroaneurysm (RAM), hypertension, retinal hemorrhage, vitreous hemorrhage, exudate, macular edema
INTRODUCTION
Retinal arterial macroaneurysms (RAMs) are acquired, focal dilations of a retinal artery, typically occurring at a bifurcation or arteriovenous crossing in the superotemporal or inferotemporal arcade before the third bifurcation of the central retinal artery.1-8 Although RAMs are usually a solitary, unilateral finding, multiple RAMs have been observed in 14-20% of cases, and bilateral disease has been observed in up to 10% of cases.1-4 Macroaneurysms are typically 100-250 microns in diameter and can be anatomically classified as fusiform or saccular.1,4,5 A fusiform RAM presents as a uniform circumferential widening of the retinal artery while a saccular RAM is a localized outpouching of the arterial wall.1,6 Based on the state of the blood-retinal barrier, RAMs can also be classified as quiescent, hemorrhagic, or exudative.4-6,9 Hemorrhagic RAMs account for approximately two-thirds of all RAMs.9
CASE REPORT
An 83-year-old white male presented for an eye examination noting a mild and gradual increase in blur at distance and near in each eye with his glasses over the last two years. His ocular history was remarkable for pseudophakia with residual astigmatism OU. His medical history included hypertension, hyperlipidemia, benign prostatic hyperplasia, erectile dysfunction, and gastroesophageal reflux disease. He had coronary artery bypass grafting done 17 years prior and had an allergy to penicillin. His medications included metoprolol succinate, chlorthalidone, simvastatin, terazosin hydrochloride, sildenafil citrate, omeprazole, and 81 mg aspirin. The patient was a former smoker but denied any history of alcohol abuse or recreational drug use.
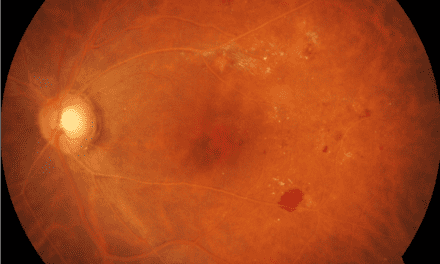
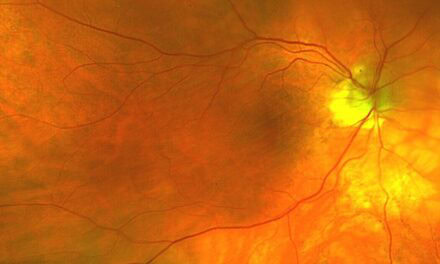
Best-corrected visual acuity was 20/25 OD and 20/30 OS. Pupils, confrontation visual fields, and extraocular muscle testing were normal. Anterior segment findings were unremarkable with a posterior chamber intraocular lens and mild posterior capsular opacification present in each eye. Intraocular pressures were 16 mmHg in each eye. Fundus examination revealed flat nerves with good color and a cup-to-disc ratio of 0.40 in the right eye and 0.35 in the left eye. A few small hard macular drusen were noted bilaterally as well as mild tortuosity and crossing changes of the blood vessels. There was no macular edema. A 1-disc diameter (DD) round, red lesion with a white center was noted along the inferotemporal arcade OD (Figure 1). The lesion was surrounded by several intraretinal hemorrhages. A 2 x 2.5 DD flat lesion with even pigmentation was noted superonasally OS (Figure 2).

Figure 1. Color and red-free photographs of the round, red lesion and surrounding hemorrhages inferotemporally OD.
Amsler grid testing was unremarkable. Optical coherence tomography (OCT) was unavailable. The patient’s blood pressure measured 134/76 mmHg. A review of his labs from two months prior indicated a normal complete blood count, comprehensive metabolic panel, and lipid profile.
The patient was diagnosed with a hemorrhagic retinal arterial macroaneurysm OD, a choroidal nevus OS, and mild nonexudative macular degeneration OU.
With regards to the macroaneursym, the patient’s primary care provider was alerted of the finding; he requested that the patient begin logging his blood pressure at home and follow-up with him in three weeks. A dilated eye exam was scheduled for three months later, and the patient was advised to call the clinic if any change was noted in the interim.
The patient presented urgently to the clinic 11 weeks later complaining of a red streak in his right eye the night before that persisted for approximately two hours. When he woke up the vision was very cloudy but seemed to slowly be clearing. He reported his blood pressure was 174/74 mmHg when his symptoms started.
Best-corrected visual acuity was 20/80 OD, with no improvement via pinhole testing. Examination findings were unchanged in both eyes, with the exception of a newly noted vitreous hemorrhage in the right eye. His blood pressure measured 152/74 mmHg. The patient was advised to avoid any strenuous activity, and keep his head elevated. A follow-up appointment with the patient’s primary care provider was arranged for the following day, and a dilated eye exam was scheduled for one week later.
The patient’s primary care provider added lisinopril to his medications, which improved his blood pressure. The patient’s vision dropped to 20/200 while the vitreous hemorrhage persisted and the intraretinal hemorrhaging worsened for two weeks, narrowly sparing the macula. The patient refused care by a retinal specialist. The vitreous hemorrhage resolved after two months. While the intraretinal hemorrhages were resolving, exudates in a circinate ring formed. These exudates resolved after two more months and the patient’s vision returned to 20/30.

Figure 3. Color and red-free photograph of the hemorrhages in the patient’s right eye 12 weeks after his initial exam.

Figure 4. Color and red-free photographs of the hemorrhages in the patient’s right eye 13 weeks after his initial exam.

Figure 5. Color photographs of the hemorrhages and exudates in the patient’s right eye 18 weeks after his initial exam.
DISCUSSION
Subretinal, intraretinal, and preretinal hemorrhages are often seen when a retinal arterial microaneurysm (RAM) ruptures because arteries are high flow vessels capable of pushing blood into multiple layers of the retina.1 In addition to retinal hemorrhages, vitreous hemorrhages have been reported in approximately 10% of cases. A recurrent vitreous hemorrhage from a single macroaneurysm is rare but may occur from a second macroaneurysm in the same eye.2 An exudative retinopathy consisting of a circinate pattern of yellow or white lipids may surround the RAM or appear in the macular region.1,2,6
RAMs are found in approximately 1 of 9000 eyes.4 They are more prevalent in individuals over 60 years old, with 60-80% of cases affecting females. Studies have found that 64-79% of patients with a RAM also have hypertension, which may play a role in its pathogenesis.1-4,6 Furthermore, it has been reported that patients with hemorrhagic RAMs tend to be more hypertensive than patients with exudative RAMs.4 Arteriosclerosis, hyperlipidemia, polycythemia, cerebrovascular disease, polyarteritis nodosa, rheumatoid arthritis, and Raynaud’s disease have also been associated with RAMs.2,3
Macular edema, with or without exudation, is considered a common early complication of RAMs, occurring in approximately one-third of reported cases.1,2,7 Epiretinal membranes, cystoid macular edema, and macular holes are considered late complications of chronic macular edema.2 One study found that macular holes developed in 5.9% of eyes with a hemorrhagic RAM and that macular holes were more common the closer a RAM was to the fovea.10 A dense preretinal or intravitreal hemorrhage may mask an underlying macular hole.4
Neurosensory retinal detachments may be seen surrounding RAMs, usually in association with edema, hemorrhage, or exudation.1,2,4 Arteriolar narrowing and irregularity, arteriovenous crossing changes, and arteriolar and venous occlusions are also frequently observed.1-4
Patients may present with an acute or insidious loss of vision when the macula is involved or when a vitreous hemorrhage is present, but often RAMs are discovered during routine examination of asymptomatic patients.1,2 Fluorescein angiography (FA) can be useful in identifying a RAM, especially if it is involuted or obscured by hemorrhages. A fusiform RAM will show rapid filling in the early arterial phase, while a saccular RAM will display minimal early filling with full filling in the middle to late phases.1,4,6 Filling in the early phases may be segmental due to clot formation or scarring.1,2,6 An involuted RAM may have a translucent shell that does not leak fluorescein.2 A Z-shaped appearance may also be appreciated at the site of a resolved RAM due to fibrotic changes in the arterial wall.6 The involved artery is commonly narrowed and irregular both proximal and distal to the aneurysm.2,6 Macular involvement may be indicated by blocked fluorescence from lipid exudates, leakage from capillaries and microaneurysms, focal or cystoid edema, or preretinal gliosis.2 In cases where fluorescein angiography is inconclusive, indocyanine green angiography (ICGA) may be helpful. RAMs appear as well-defined areas of hyperfluorescence with ICGA.1
Optical coherence tomography (OCT) may also be beneficial in diagnosing RAMs and in monitoring associated subretinal fluid, macular edema, and hemorrhaging. An OCT scan directed through a RAM shows an abnormal expansion of the blood vessel wall and an enlarged lumen located in the inner retinal layers. The RAM elevates the internal limiting membrane and ganglion cell layers and shadows deeper retinal layers.1 OCT angiography (OCTA) can provide information about blood flow, including presence or absence of a thrombus, which can aid in treatment.11,12 While OCT is a less invasive procedure, its use may be limited by a dense preretinal or vitreous hemorrhage.1
The pathophysiology of RAMs is not fully understood, but it has been hypothesized that a RAM results from a focal weakness in the arterial wall, which may have resulted from an embolus or thrombus, or may simply be due to aging and arteriosclerosis.1,2 Aging causes atrophy of the muscularis layer, which leads to thinning and fibrosis of the vessel wall and ultimately decreased elasticity of the artery. The lack of elasticity leaves the arterial wall susceptible to dilation from elevated hydrostatic pressure, as is seen in hypertension.1-4,6 In addition, it has been found that the absence of the adventitial layer at arteriovenous crossings makes these areas more prone to aneurysm formation.1,4,6,8
Long-term visual prognosis is generally good but depends on the location of the RAM and associated retinal changes as well as the severity and duration of the disease.1,2,4,6 While most RAMs follow a benign course of thrombosis, fibrosis, and spontaneous resolution, a permanent reduction of best corrected vision may occur secondary to hemorrhages, exudates, subretinal neovascularization, an epiretinal membrane, chronic macular edema, a macular hole, or a neurosensory retinal detachment.1,2,4,5,9 Hemorrhagic RAMs have a better visual prognosis than exudative RAMs.4,6,9 Eyes that had a vitreous or preretinal hemorrhage tend to have a better final visual acuity than eyes that had a subretinal hemorrhage.1,4,6,8 Submacular hemorrhaging lends itself to the poorest visual outcome due to permanent damage to the retinal pigment epithelium and photoreceptors. Chronic macular edema and macular exudates are the most common cause of poor visual outcome in patients with RAMs.1,2,4,8
Treatment and management of a RAM depends on the clinical appearance and associated complications. Meta analysis by Chen et al found that 64% of RAMs resolved without intervention.9 Hemorrhagic RAMs without macular involvement should be monitored every 1-3 months while asymptomatic RAMs without macular involvement can be observed every 3-6 months.1,2 Patients with reduced vision secondary to retinal or vitreous hemorrhage, without significant edema or exudation, should be closely observed for this first 3-4 months due to the high frequency of spontaneous resolution and limited sequalae from these findings.1,2,6,8 A recurrent vitreous hemorrhage secondary to the same macroaneurysm is rare, but if it occurs this may warrant laser photocoagulation.2 In cases of direct macular involvement by edema, exudation, subretinal hemorrhaging, or sensory detachment, referral to a retina specialist is indicated for treatment.1,2,4,7,8
Photocoagulation may be applied directly to a RAM with the intent to speed involution and decrease leakage without occluding the artery itself.1,2,5,13 Indirect photocoagulation destroys some of the adjacent retina supplied by the artery, thereby decreasing the need for arterial blood, reducing blood flow and the intraluminal pressure in the artery, thus facilitating resolution of the macroaneurysm.2 Theoretically, indirect treatment avoids the risk of rupture and hemorrhage; however, these complications have not been reported in conjunction with direct photocoagulation.2,13 Complications of direct and indirect laser photocoagulation include arteriolar occlusion, formation of an arteriovenous shunt, formation of a secondary macroaneurysm, inadvertent foveal or optic nerve photocoagulation, increased exudation, retinal or vitreous hemorrhage, choroidal neovascularization, subretinal fibrosis and scarring, and epiretinal membrane formation.1,2,4,8,13 To avoid these complications, subthreshold laser treatment (STLT), which utilizes a shortened laser exposure time and subvisible clinical endpoint, may be performed.1,14 A study by Battaglia et al found that both threshold laser treatment (TLT) and STLT achieved similar improvements in visual acuity. Atrophic scars only surrounded RAMs that were treated with TLT; no complications were appreciated in those treated with STLT.14 Chen et al found that 96% of RAMs treated with laser photocoagulation resolved, and 73% had visual improvement of one line or more suggesting treatment with last may be more beneficial than observation.9
Intravitreal injections of anti-vascular endothelial growth factor (VEGF) have been studied as off-label treatment options for RAMs with promising results.7,9 Most of the reported cases have shown macroaneurysm or macular edema regression after two or three intravitreal injections of bevacizumab or ranibizumab, however, chronic cases may take longer.1,5,7,8,15,16 Chen et al found a 98% RAM closure rate in patients treated with anti-VEGF, which was better than the 73% RAM closure rate found in patients treated with photocoagulation. Likewise, visual acuities improved by more than 1 line in 90% of patients treated with anti-VEGF compared to 73% of the patients treated with photocoagulation.9 Potential complications of anti-VEGF injections include endophthalmitis, arterial thrombotic events, retinal tears, retinal detachment, vitreous hemorrhage, ocular hypertension, and cataract.8,17
In cases where treatment with laser photocoagulation or anti-VEGF therapy alone is not effective, a combination of the two treatments may be warranted, particularly if there is persistent macular exudation. Chen et al found that both hemorrhagic and exudative RAMs responded well to combination treatments. Hemorrhagic RAMs generally respond better to laser treatments while exudative RAMs respond better to anti-VEGF treatments.9
A pars plana vitrectomy (PPV) may be warranted in cases of dense or non-clearing vitreous hemorrhage to allow a clinician to subsequently view and manage other retinal complications.4,6 A PPV may also be performed to prevent epiretinal membrane formation and toxic effects on the retinal tissue due to the sustained presence of dense preretinal blood.1,2 Potential complications of a PPV include post-operative cataract progression, a retinal break with or without retinal detachment, macular hole formation, recurrent bleeding from the RAM, proliferative vitreoretinopathy, and endophthalmitis.1
A preretinal hemorrhage may be treated by a pulsed neodymium;yttrium aluminum garnet (Nd:YAG) laser membranotomy to speed visual recovery, decrease complications of stagnant preretinal blood, and increase visualization of the retina. This causes a focal disruption of the inner limiting membrane of the retina, allowing the hemorrhage to dissipate into the vitreous for quicker reabsorption. Possible complications of this procedure include a secondary non-clearing vitreous hemorrhage, retinal detachment, macular hole, or epiretinal membrane.1
A submacular hemorrhage can be treated with pneumatic displacement or submacular surgery. Pneumatic displacement uses injected perfluorocarbon gas and prone positioning to displace the hemorrhage away from the macula, thereby reducing complications in the macula from the presence of subretinal blood. Submacular surgery includes a PPV followed by surgical drainage of submacular blood via a retinotomy, fluid-air exchange, and prone positioning for one week. Possible complications of these two procedures include cataract progression and a retinal break with or without detachment. Some speculate complications with pneumatic displacement should be less frequent than with submacular surgery.1
Conclusion
Retinal arterial macroaneurysms (RAMs) present as either a uniform widening of the retinal artery or a localized outpouching of the arterial wall. Fluorescein angiography, indocyanine green angiography, optical coherence tomography, and optical coherence angiography can be useful in identifying a RAM. While most RAMs have a benign course with spontaneous resolution, associated retinal changes may cause a reduction in vision and warrant treatment via direct or indirect photocoagulation, intravitreal injections of anti-vascular endothelial growth factor, a pars plana vitrectomy, a laser membranotomy, pneumatic displacement, or submacular surgery. Patients with a RAM should also be evaluated by a primary care physician for hypertension as a strong correlation exists between the two conditions.
REFERENCES
- Speilburg AM, Klemencic SA. Ruptured retinal arterial macroaneurysm: diagnosis and management. J Optom 2013;7(3):131-7.
- Rabb MF, Gagliano DA, Teske MP. Retinal arterial macroaneurysms. Surv Ophthal 1988 Sept-Oct;33(2):73-96.
- Panton RW, Goldberg MF, Farber MD. Retinal arterial macroaneurysms: risk factors and natural history. Br J Ophthalmol 1990;74:595–600.
- Pitkanen L, Tommila P, Kaarniranta K, Jaaskelainen JE, Kinnunen K. Retinal arterial macroaneurysms. Acta Ophthamol 2014;92:101-4.
- Menezes C, Carvalho R, Teixeira C, Lemos JA, Goncalves R, Coelho P, Lima A. Foveal exudative macroaneurysm treated with intravitreal ranibizumab. Case Rep Ophthalmol 2015 May-Aug;6(2):170-5.
- Moosavi RA, Fong KC, Chopdar A. Retinal artery macroaneurysms: clinical and fluorescein angiographic features in 34 patients. Eye 2006;20:1011–20.
- Leung EH, Reddy AK, Vedula AS, Flynn HW. Serial bevacizumab injections and laser photocoagulation for macular edema associated with a retinal artery macroaneurysm. Clin Ophthalmol 2015;9:601-9.
- Erol MK, Dogan B, Coban DT, Toslak D, Cengiz A, Ozel D. Intravitreal ranibizumab therapy for retinal arterial macroaneurysm. Int J Clin Exp Med 2015;8(7):11572-8.
- Chen H, Zhao X, Meng L, Zhang W, Chen Y. Clinical characteristics of retinal arterial macroaneurysms and prognosis of different interventions. Graefes Arch Clin Exp Ophthalmol 2022;260:439-450.
- Tashimo A, Mitamura Y, Ohtsuka K, Okushiba U, Imizumi H, Takeda M. Macular hole formation following ruptured retinal arterial macroaneurysm. Am J Ophthalmol 2003 Apr;135(4):487-92.
- Astroz P, Miere A, Cohen SY, Querques G, Souied EH. Optical coherence tomography angiography in the diagnosis and follow-up of retinal arterial macroaneurysms. Retinal Cases & Brief Reports 2021;16:1-4.
- Miura M, Muramatsu D, Hong Y-J, Yasuno Y, Itami A, Iwasaki T, Goto H. Noninvasive vascular imaging of ruptured retinal arterial macroaneurysms by doppler optical coherence tomography. BCM Ophthalmol 2015;15:79.
- Brown DM, Sobol WM, Folk JC, Weingeist TA. Retinal arteriolar macroaneurysms: long-term visual outcome. Br J Ophthalmol 1994;78:534–8.
- Battaglia Parodi M, Iacono P, Pierro L, Papayannis A, Kontadakis S, Bandello FM. Subthreshold laser treatment versus threshold laser treatment for symptomatic retinal arterial macroaneurysm. Invest Ophthalmol Vis Sci 2012;53:1783–6.
- Jonas JB, Schmidbauer M. Intravitreal bevacizumab for retinal macroaneurysm. Acta Ophthalmol 2010;88:e284–3768.
- Wenkstern AR, Petersen H. Intravitreal ranibizumab in retinal macroaneurysm. Graefe’s Arch Clin Exp Ophthalmol 2010 Nov;248(11):1667-70.
- Moshfeghi AA. Safety of intravitreal anti-VEGF agents. Rev Ophthal 2014 Nov.
Dr. Knapke graduated from The Ohio State University College of Optometry in 2010. She completed a primary care residency at the Dayton VA Medical Center in 2010-2011 and subsequently became a staff optometrist at their Lima Community Based Outpatient Clinic.