Highlighting hypertriglyceridemia with a case of lipemia retinalis

ABSTRACT
BACKGROUND
Severe hypertriglyceridemia can transiently cause the appearance of a salmon-colored fundus with creamy vessels which is known as lipemia retinalis.1 This case presents images taken during and after an episode of lipemia retinalis including specific OCT findings. This case will review pertinent lipid panel findings, systemic management for hypertriglyceridemia, and the relationship between hypertriglyceridemia and diabetes mellitus.
CASE REPORT
A 42-year-old female with uncontrolled diabetes mellitus and hypertriglyceridemia presented for an annual eye exam. She reported poor adherence to her prescribed medications and had been lost to follow-up with her primary care physician. On eye exam, her best corrected vision was mildly decreased in both eyes with bilateral moderate nonproliferative diabetic retinopathy. In addition, her fundus was lighter in color and all retinal vessels appeared creamy and attenuated bilaterally. With the diagnosis of lipemia retinalis, she was referred back to her primary care physician where her triglyceride level was found to be dramatically elevated. She restarted her prescribed fibrate medication and within 2 weeks her triglyceride levels were trending towards normal and the lipemia retinalis had fully resolved.
CONCLUSION
The rare condition of lipemia retinalis is the result of extremely elevated triglyceride levels.1 Retinal findings resolve with the initiation of appropriate systemic treatment. Understanding the systemic treatment and co-management with a patient’s medical providers isis crucial in preventing life-threatening complications.1-2
Keywords: hypertriglyceridemia, lipemia retinalis, lipid panel, fibrate medications
INTRODUCTION
Severely high triglyceride levels can lead to the ocular condition lipemia retinalis.1-2 Lipemia retinalis results in a salmon-colored fundus and creamy retinal vessels. This is a marker for eye care providers to recognize elevated and dangerous triglyceride levels in a patient, often from an uncontrolled secondary metabolic disorder.3 Lipemia retinalis is not sight-threatening and it is transient with appropriate systemic management.1-2 Here, a case of lipemia retinalis in a diabetic patient is described which includes OCT imaging, a lipid panel review, and an overview of both pharmacologic and nonpharmacologic treatment recommendations.
CASE REPORT
A 42-year-old female presented for a comprehensive eye exam. Her previous eye exam was two years prior and was remarkable for a mild hyperopic refractive error and moderate nonproliferative diabetic retinopathy (NPDR) without clinically significant macular edema (CSME) in both eyes. Her systemic history was remarkable for type 2 diabetes mellitus that was diagnosed 12 years prior and was managed with metformin and insulin. In addition, she carried a diagnosis of hypertension (treated with lisinopril), hypertriglyceridemia (treated with fenofibrate), genital herpes, and depression. Her social determinants of health included homelessness and food insecurity. The patient was often lost to follow-up. Medication adherence had been challenging for the patient and she had periods of extended medication non-adherence. During this exam she reported that she was not taking fenofibrate and admitted to part-time adherence to her other listed medications.
Best corrected visual acuity was 20/25 at distance and near in the right and left eye with a small increase in the hyperopic prescription. Entrance testing including pupils, extraocular muscles, and confrontation fields was unremarkable in both eyes. The anterior segment was unremarkable without neovascularization of the iris or notable lens changes. Intraocular pressure was 18mmHg in each eye by Goldmann applanation tonometry.
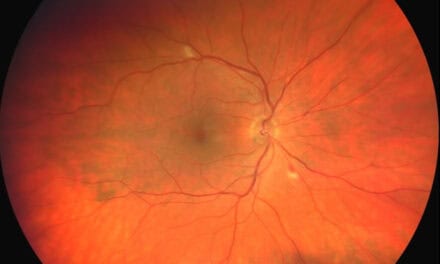
On dilated exam, intraretinal hemorrhages, cotton wool spots, and exudation were present temporally to the macula in both eyes, worse in the left eye. While there wasn’t obvious edema involving the fovea in either eye, the exudation and hemorrhaging were within 500 microns of the foveal center. Both retinas exhibited a much lighter “salmon-colored” appearance than previously documented. Both the arteries and veins appeared creamy throughout the entire posterior pole and periphery. The vessels were of normal course, but there was mild arteriolar attenuation. The optic nerves appeared pink and healthy without edema or neovascularization. The peripheral retina was unremarkable in both eyes. Fundus photography and OCT images were acquired that day and are shown in Figures 1-2.

Figure 1. Fundus photography of both eyes. The retina appears much lighter in color and both arteries and veins appear creamy. Intraretinal hemorrhages, cotton wool spots, and exudation are present temporally to the macula in both eyes. The left eye has notably worse retinopathy with CSME.

Figure 2. A cross section from the Cirrus Macular Cube 512×128 OD and OS. The cross section highlights the vessel changes that can be seen on the OCT from lipemia retinalis. Vessels appear very hyperreflective and mildly engorged (yellow arrows). Hyperreflective dots within the inner nuclear layer are likely lipid leakage (red arrow),13 whereas irregular and hyperreflective changes in the outer nuclear and outer plexiform layers represent hemorrhages and exudation from the diabetic retinopathy (best seen temporally OS).
After the dilated exam, the patient’s primary care physician was notified of the exam findings and a lipid panel was ordered. Her physician restarted her on her prescribed fenofibrate 160 mg daily. One week later she had her lipid panel drawn and read (Figure 3). Her triglyceride level was 1,139 mg/dL (normal range: 0-150 mg/dL). This level was drawn one week after restarting fenofibrate, therefore, her triglyceride level was likely even higher on initial presentation.

Figure 3. Results of the lipid panel drawn within one week of the presentation of lipemia retinalis.
Within two weeks of her eye exam, the patient had a telemedicine visit with both her primary care physician and her endocrinologist to review medication adherence and labs. No changes were made to her medication regimen, as both providers believed that with consistent adherence, her systemic profile would improve drastically. She was seen in the eye clinic again three months after the initial appointment. At this follow up, the moderate NPDR with CSME had mildly improved in each eye. Her best corrected vision remained 20/25 in the right and left eye. However, her lipemia retinalis had completely resolved and her fundus coloring and vessel appearance returned to baseline (Figure 4). Another lipid panel was drawn and read after this appointment and after three months of consistent fibrate medication adherence, the patient’s triglyceride level was down to 310 mg/dL.

Figure 4. Fundus photography of both eyes three months after the initial presentation. The retina and retinal vasculature color appear back to baseline in both eyes. Both eyes still have few scattered hemes and microaneurysms and cotton wool spots, although improved.
DISCUSSION
Hypertriglyceridemia can occur as a primary familial disorder or can be secondary to other metabolic diseases, most commonly uncontrolled hypertension or diabetes mellitus.3 Any age group can be affected, from premature infants to the elderly. Hypertriglyceridemia predominantly affects males and is most likely to occur between the ages 40-59.3 Lipemia retinalis is a visible ophthalmic manifestation of severe hypertriglyceridemia, which does not often result in ocular symptoms but can hint at a more serious complication or an uncontrolled primary disorder.2
Hypertriglyceridemia is defined as serum triglycerides above 150 mg/dL. The Endocrine Society defines levels from mild to very severe hypertriglyceridemia (Table 1).4 Lipemia retinalis occurs when levels are extremely elevated (>2500 mg/dL).1,3 The retinal vasculature changes start in the periphery and as triglyceride levels increase, the vascular changes are seen within the posterior pole. In the final stage, the fundus presents with a “salmon-color.” There are three stages of lipemia retinalis that link triglyceride level with the extent of fundoscopic findings (Table 2).1,3 While the triglyceride level at the exact time of presentation is unknown since the patient had re-started her fibrate medication after the eye exam and the lipid panel was drawn one week later. Therefore, the level of 1,139 mg/dL doesn’t represent her presenting triglyceride level. The presence of posterior pole involvement is suggestive of triglyceride levels consistent with grade 3 lipemia retinalis, which may have been over 5000 mg/dL (Table 2).
The pathophysiology of lipemia retinalis is based on the primary triglyceride carrying molecule, the chylomicron.5,9 Chylomicrons are large lipid transport molecules that carry digested triglycerides from the intestines through systemic circulation.9 A high level of triglycerides in chylomicrons causes light to scatter and results in the creamy vessel appearance of lipemia retinalis.1,5 A standard lipid panel order includes a measure of triglyceride levels along with total cholesterol and the cholesterol lipoprotein carrier molecules: high-density lipoprotein (HDL), low-density lipoprotein (LDL), and sometimes very-low-density lipoprotein (VLDL). Triglyceride levels are used to calculate LDL. High levels of LDL put patients at risk for heart disease and stroke.6-7
As mentioned above, hypertriglyceridemia is often asymptomatic. However, in addition to lipemia retinalis, patients may experience eruptive xanthomas, abdominal pain, peripheral neuropathy, or memory loss. Acute pancreatitis is the most life-threatening complication of hypertriglyceridemia.2 Familial disorders, including apolipoprotein C-II (ApoC-II) deficiency and lipoprotein lipase (LPL) deficiency, lead to chylomicronemia. In these presentations, triglyceride levels exceed 1000 mg/dL.2,7 In this case, the patient’s elevated triglyceride levels were most likely due to her concurrent conditions of uncontrolled diabetes and hypertension, the two most common causes of secondary hypertriglyceridemia. Obesity, renal failure, and hypothyroidism are other common primary metabolic disorders that may present with a secondary hypertriglyceridemia.1-2,7-8
Additional ocular manifestations of severe hypertriglyceridemia have been documented in case reports, though they are rare. These include xanthomas of the periocular area, conjunctiva, iris, retina, and choroid, as well as venous stasis and red blood cell aggregation secondary to hyperchylomicronemia resulting in venous and arterial occlusions.5,10,11-12 In patients with lipemia retinalis, spectral-domain optical coherence tomography (SD-OCT) reveals hyperreflective and engorged retinal vessels, as well as hyperreflective dots that accumulate in the inner nuclear layer of the retina, representing lipid leakage from retinal capillaries.13 Both of these findings are evident in this case (Figure 2). Neither fluorescein angiography nor OCTA have been shown to be sensitive to changes in lipemia retinalis.14
Lipemia retinalis requires no direct treatment to the eye; management is specifically focused on systemic control of triglyceride levels. As triglyceride levels improve, so will the retinal appearance. First line treatment is strict adherence to a low-fat diet and an increase in physical activity. This includes decreasing carbohydrates and increasing dietary fiber.2 Strict glycemic control is also encouraged in those with uncontrolled diabetes mellitus.15 Fibric acid derivatives (fibrates) are the first-line triglyceride lowering medication.16 While statin medications are utilized to decrease LDL levels, they have little or no effect on triglyceride levels, so in these presentations, fibrates are recommended.15-16 In patients not reaching LDL or non-HDL goals, fibrates can be used as a adjunct therapy, however, there is risk of myopathy or rhabdomyolysis in combination therapy.16 Both statins and fibrates are safe and well-tolerated medications and aid in the reduction of cardiovascular events.16 In this case, when the patient restarted her fibrate medication, there was a rapid and drastic improvement in both her measured triglyceride levels and fundoscopic manifestations.
CONCLUSION
This case reinforces the importance of systemic management of hypertriglyceridemia. The ocular presentation of lipemia retinalis doesn’t represent a sight-threatening emergency, but it gives insight into a potential systemic emergency. Simply by improving medication adherence in this case, the patient was able to decrease her triglyceride level significantly and resolve the lipemia retinalis. Severe hypertriglyceridemia is life-threatening and an optometrist’s role in recognizing the retinal signs is crucial to management of the condition.
REFERENCES
- Zahavi A, Snir M, Kella YR. Lipemia retinalis: case report and review of the literature. J AAPOS. 2013 Feb;17(1):110-1. doi: 10.1016/j.jaapos.2012.10.010. Epub 2013 Jan 18. PMID: 23337350.
- Horton M, Thompson K. Lipemia retinalis preceding acute pancreatitis. Optometry. 2011 Aug;82(8):475-80. doi: 10.1016/j.optm.2011.01.011. Epub 2011 May 12. PMID: 21570360.
- Christian JB, Bourgeois N, Snipes R, Lowe KA. Prevalence of severe (500 to 2,000 mg/dl) hypertriglyceridemia in United States adults. Am J Cardiol. 2011 Mar 15;107(6):891-7
- Berglund L, Brunzell JD, Goldberg AC, et al. Evaluation and treatment of hypertriglyceridemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2012; 97:2969.
- Vinger PF, Sachs BA. Ocular manifestations of hyperlipoproteinemia. Am J Ophthalmol. 1970 Oct;70(4):563-73
- Grundy SM, Stone NJ, Bailey AL, et al.Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019; 139:e1082.
- Santamarina-Fojo S. The familial chylomicronemia syndrome. Endocrinol Metab Clin North Am 1998; 27:551.
- Goldberg IJ. Hypertriglyceridemia: impact and treatment. Endocrinol Metab Clin North Am. 2009 Mar;38(1):137-49. doi: 10.1016/j.ecl.2008.11.005. PMID: 19217516.
- Rayner S, Lee N, Leslie D, Thompson G. Lipaemia retinalis: a question of chylomicrons? Eye (Lond). 1996;10 ( Pt 5):603-8.
- Gopal L, Sunder KS, Rao SK, Soni M, Sharma S, Ramakrishnan S. Hyperlipidemia in a poorly controlled diabetic presenting with lipemic aqueous and lipemia retinalis. Retina (Philadelphia, Pa.). 2004 Apr;24(2):312-5.
- P.K. Nagra, A.C. Ho, J.D. Dugan Jr. Lipemia retinalis associated with branch retinal vein occlusion. Am J Ophthalmol, 135 (2003), pp. 539-542.
- Zheng C, Lin Y, Jiang B, Zhu X, Lin Q, Luo W, Tang M, Xie L. Plasma lipid levels and risk of retinal vascular occlusion: A genetic study using Mendelian randomization. Front Endocrinol (Lausanne). 2022 Oct 10;13:954453. doi: 10.3389/fendo.2022.954453. PMID: 36299452; PMCID: PMC9588969.
- Özturk BT, Bozkurt B, Meşen A, Gonul S, İpekci SH. Spectral-Domain Optical Coherence Tomography Findings in Lipemia Retinalis. Ophthalmic Surg Lasers Imaging Retina. 2016 Jun 1;47(6):589-92. doi: 10.3928/23258160-20160601-13. PMID: 27327291.
- Zhang SJ, Yan ZY, Yuan LF, Wang YH, Wang LF. Multimodal imaging study of lipemia retinalis with diabetic retinopathy: A case report. World J Clin Cases. 2022 Jul 6;10(19):6736-6743. doi: 10.12998/wjcc.v10.i19.6736. PMID: 35979318; PMCID: PMC9294873.
- Leaf DA. Chylomicronemia and the chylomicronemia syndrome: a practical approach to management. Am. J. Med. 2008 Jan;121(1):10-2.
- Jacobson TA, Zimmerman FH. Fibrates in combination with statins in the management of dyslipidemia. J Clin Hypertens (Greenwich). 2006 Jan;8(1):35-41; quiz 42-3. doi: 10.1111/j.1524-6175.2005.05278.x. PMID: 16407687; PMCID: PMC8112375.