Primary central nervous system diffuse large B-cell lymphoma presenting as homonymous hemianopia

ABSTRACT
Background:
There are several factors that increase the risk of lymphoma, such as increased age, impaired immunity or infections such as the Epstein-Barr virus. Post-transplant lymphoproliferative disorder (PTLD) can develop from suppressed immunity secondary to organ transplantation. The most significant risk factor for developing PTLD is the presence of Epstein-Barr virus. PTLD is diagnosed through different testing methods including laboratory studies, radiologic imaging and pathology results. Primary central nervous system lymphoma occurs when the first organ that develops lymphoma is the central nervous system. When the lesion affects the visual pathway, visual field deficits can occur and have visually significant consequences.
Case report:
A 66-year-old male presented to the eye clinic for an eye exam with binocular vision complaints and subacute headaches. Visual field testing confirmed a left absolute homonymous hemianopia. Magnetic resonance imaging of the brain was notable for a right occipital lobe mass without herniation or hydrocephalus with a separate lesion in the left temporal horn without mass effect. The right occipital lobe mass was biopsied and positive for a diffuse large B-cell lymphoma and Epstein-Barr virus. The etiology of the primary central nervous system lymphoma was due to post-transplant lymphoproliferative disorder from the liver transplant that the patient received eight years prior. The patient’s treatment plan included chemotherapy, whole-brain radiotherapy and mass resection via craniotomy. He had multiple visits with the acquired brain injury clinic in optometry to assist with his binocular vision concerns and left visual field loss.
Conclusion:
Post-transplant lymphoproliferative disorder rarely affects the central nervous system although it can have visually significant consequences depending on the location of intracranial involvement. Patients need to undergo routine & supplemental optometric testing and consider an expeditious referral to the next level of care when neurological findings are noted on examination. Ocular rehabilitation techniques can also improve the quality of life in patients with acquired brain injury. A multidisciplinary care team is needed to meet the patient’s needs, as no standardized treatment protocols exist.
Keywords: diffuse large B-cell lymphoma, homonymous hemianopia, post-transplant lymphoproliferative disorder
INTRODUCTION
Intracranial tumors within the occipital lobe are a known cause of homonymous hemianopia. Primary central nervous system diffuse large B-cell lymphoma (DLBCL) secondary to post-transplant lymphoproliferative disorder (PTLD) is a rare type of intracranial tumor, making it an uncommon etiology of this field defect.1,2,3 The risk of developing PTLD over a lifetime varies based on the type of organ that was transplanted.4 There are multiple factors that are critical in the assessment and management of DLBCL, such as the presence of Epstein-Barr virus and chronic immunosuppression.2,3,5 A patient with PTLD that carries the Epstein-Barr virus is at a significantly higher risk of developing lymphoma shortly after an organ transplant.2,3 Rarely, an Epstein-Barr virus-positive patient can be diagnosed with PTLD years after solid organ transplantation.2 Several treatment options have shown regression of DLBCL; however, there are no definitive management guidelines.2,5
CASE REPORT
A 66-year-old Caucasian male presented to the eye clinic complaining of difficulty tracking while reading, changes in depth perception and headaches ongoing for one month. The patient also reported that he was in a car accident where he drove his car onto a sidewalk during the same timeframe. His medical history was positive for chronic kidney disease stage III, coronary artery disease, hepatitis C virus, end-stage liver disease with a history of a liver transplant eight years prior which was managed with prednisone, and chronic hypoglobulinemia and anemia secondary to his liver transplant. Best-corrected visual acuity was 20/30 in the right eye and 20/40 in the left eye. Confrontation visual field testing was positive for a left-sided defect in each eye. Intraocular pressures were normal. Anterior segment examination revealed nuclear sclerosis and posterior subcapsular cataracts in each lens, consistent with long-term prednisone use. Posterior segment examination revealed several small intra-retinal hemorrhages in both eyes. A perimetry visual field 30-2 test confirmed a left absolute homonymous hemianopia with symmetrical crossing over the vertical midline into the right superior field of both eyes (Figure 1). Neurological testing showed intact cranial nerves III-XII and fluent speech. The patient was escorted to the emergency department due to the presence of sub-acute neurologic findings.

Figure 1. 30-2 threshold visual field test of the right and left eye shows a left homonymous hemianopia with extension into the right superior quadrants.
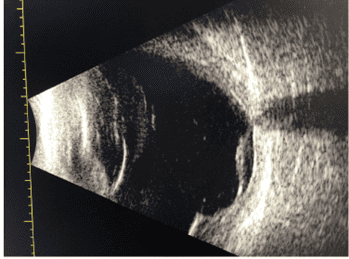
The patient underwent stat laboratory testing and neuroimaging. Laboratory testing showed decreased red blood cells, hemoglobin and hematocrit consistent with anemia, an elevated erythromycin sedimentation rate, an elevated creatinine level and reduced glomerular filtration rate consistent with kidney disease. Magnetic resonance imaging of the brain was notable for a right occipital lobe mass without herniation or hydrocephalus with a separate lesion in the left temporal horn without mass effect (Figure 2). Computed tomography scans of the brain also revealed a two-to-three-millimeter left midline shift. Computed tomography scans of the abdomen, pelvis and chest were unremarkable. Neurology was consulted and determined that a biopsy of the cranial mass was warranted to determine its pathology.

Figure 2. Magnetic resonance imaging shows a hyperreflective mass in the right occipital lobe which was biopsied to reveal a primary central nervous system lymphoma.
Within one week of his initial presentation to the eye clinic, the patient underwent neurosurgery to biopsy and resect a portion of the lesion in the occipital lobe that was immediately available during the procedure. The pathology result was positive for a diffuse large B-cell lymphoma and Epstein-Barr virus. The etiology of the primary central nervous system lymphoma was due to post-transplant lymphoproliferative disorder from the liver transplant that the patient received eight years prior. The patient was referred to oncology who initiated treatment with 8000 mg per m2 of intravenous high-dose methotrexate and 375 mg per m2 of intravenous rituximab. The dosage with this combination therapy was slowly reduced while monitoring his hepatic and renal function closely. He underwent eight cycles of methotrexate and rituximab, which had mixed results. Magnetic resonance imaging of the brain showed a persistent lesion in the right occipital lobe. Further chemotherapy was not advised due to chemotherapy toxicity and decreasing functional status. Whole brain radiotherapy was recommended and initiated. A slight decrease in the size of the right occipital mass was noted three months after the completion of whole brain irradiation. The decision was made to monitor without further treatment due to stability and limited treatment options. The patient continued headache pain management with oxycodone.
The patient returned to the acquired brain injury clinic for evaluation one month after his initial visit to the eye clinic. His testing was positive for convergence insufficiency, mild left neglect, and stable homonymous hemianopia. Press-on Peli-prisms were trialed to help compensate for the left visual field loss. The initial prism trial was successful; however, on subsequent visits he could not remember to use the prism and the press on prisms were discontinued. It is important to note that the patient’s inability to use the prism may be a function of his visual neglect. Following evaluation with the optometry brain injury clinic, the patient was referred for vision rehabilitation, including vision therapy and orientation and mobility training. The vision therapy plans included exercises to improve positive fusional vergences and vergence facility, pursuit and saccadic movements, and scanning into the blind hemifield. Exercises recommended in the patient’s therapy plan include Hart Chart Saccades, Michigan Tracking, Marsden Ball and Rotating Peg Board for oculomotor function. Yoked prism and MacDonald Chart were recommended for peripheral awareness and training on the Bioness Integrated Training System, which combines exercises for vision, motor, balance and cognition. Vergence training was initiated and slowly progressed as the patient was able. Vergence exercises included Brock String, Vectograms, Prism Flippers, Lifesaver cards, and Aperture Ruler. Orientation and mobility training was provided by a blind rehabilitation specialist (BRS) to ensure the safety of the patient when moving in the environment with his visual field loss. Following vision rehabilitation, the patient continued to struggle with reading due to the left hemianopia and residual convergence insufficiency. At discharge from vision rehabilitation services, the BRS further trained the patient on the use of a hemianopia reading guide and provided an audible medicine reader to aid in his medication management. The patient continued care in the eye clinic to assess his ocular health and the stability of the visual field defect.
The patient returned to the neurosurgery clinic nine months after radiotherapy with worsening headaches despite a stable occipital lobe lesion on MRI. The decision was made to remove a small lesion that remained in the right occipital region to determine if this would improve his headaches. The patient underwent the reresection via craniotomy, and the mass was removed altogether. He reported improvement in his headaches at his two weeks post-operative visit. The pathology results showed no residual lymphoma. Magnetic resonance imaging one year following the second resection shows the surgical cavity with post-craniotomy encephalomalacia of the right occipital lobe. (Figure 3).

Figure 3. Magnetic resonance imaging one year following the second resection shows the surgical cavity with postcraniotomy encephalomalacia of the right occipital lobe with a coronal, axial and sagittal cut respectively.
He has been on surveillance for over two years without evidence of disease recurrence. A perimetry visual field 24-2 test was completed, revealing a stable left absolute hemianopia (Figure 4). The defect crossing the vertical midline into the right superior field was not noted. The perimetry visual field 24-2 test was chosen over the 30-2 test due to the shorter test duration and to minimize discomfort for the patient. In an ideal situation, a 30-2 would have been selected in the presence of neurological disease to allow for a direct comparison of pre-surgical and post-surgical visual field defects. He continues to be monitored by oncology every six months with a brain MRI and labs.

Figure 4. 24-2 threshold visual field test of the right and left eye shows a left homonymous hemianopia two years following the second resection.
DISCUSSION
Lymphomas develop when a state of immunodeficiency is present, such as when carrying the human immunodeficiency virus, using immunosuppressants for autoimmune disease or after an organ transplant.6 Organ transplant patients depend on immunosuppressant medications, such as prednisone, mycophenolate mofetil, tacrolimus, cyclosporine and azathioprine, to prevent rejection.3 The immunosuppression achieved by these medications increases the likelihood of illness from viruses, bacteria and cancer.2 When lymphoma occurs due to suppressed immunity secondary to organ transplantation, it is called post-transplant lymphoproliferative disorder (PTLD).2 In this case, his liver transplant and immunosuppression from chronic prednisone use predisposed the patient to develop PTLD. There is a less than 10 percent risk of developing PTLD after liver transplantation.4 When the first organ system to develop lymphoma is the central nervous system, it is termed primary central nervous system lymphoma, a type of non-Hodgkin’s lymphoma.6 Primary central nervous system lymphoma represents approximately two percent of all primary central nervous system tumors.5 The most common presenting symptom of patients with primary central nervous system PTLD is a headache, which was present in this case.7
There are four forms of post-transplant lymphoproliferative disorder lymphomas: early type, polymorphic, monomorphic, and classic Hodgkin-type lymphoma.2,3 Monomorphic lymphoma is the most common type, which can involve B-cells or T-cells, with B-cell being the most prevalent.2,3 Diffuse large B-cell lymphoma (DLBCL) is the most common of monomorphic PTLD types involving B-cells.2,3,5 Other types of B-cell lymphoma from PTLD include Burkitt lymphoma, plasma cell myeloma, and plasmablastic lymphoma.2,5 The prevalence of DLBCL is seven per 100,000 people in the United States, and only 20 percent of all DLBCL occur after solid organ transplant.1,2 Primary involvement of the central nervous system alone is rare, only occurring in seven percent of all PTLD cases.3 Typically, the central nervous system is involved secondary to a B-cell lymphoma in other organ systems.3,5
Risk factors for the development of PTLD include the presence of Epstein-Barr virus and chronic immunosuppression.2,3,6 Epstein-Barr virus, which occurs in 90 percent of the population worldwide, is the most significant risk factor for developing PTLD in patients with solid organ transplantation.3,8 Epstein-Barr virus invades and immortalizes B-cells, contributing to tumor cell formation.2,3 Immunocompromised patients cannot activate cytotoxic T-cell response due to immune suppression to fight the cancerous cells, which allows the indestructible B-cells to proliferate into a tumorous mass.2,3 When Epstein-Barr virus is absent, there is a 0.2% chance of developing PTLD.2 Epstein-Barr virus-negative PTLD typically occurs seven to ten years after organ transplantation, whereas 90% of Epstein-Barr virus-positive PTLD usually occurs within months of transplantation.2 Interestingly enough, manifestations of PTLD, in this case, did not occur until eight years post-transplant, which is atypical given he was Epstein-Barr virus-positive.
PTLD is diagnosed through laboratory and Epstein-Barr virus testing, radiologic imaging, and pathologic studies. Primary central nervous system lymphoma is multifocal in 37-61% of cases, as demonstrated in this case with the presence of a right occipital lobe mass and left temporal horn lesion noted on neuroimaging.5,6 Primary central nervous system lymphoma is localized within the cerebral hemispheres in 85% of cases.6 The location of a lesion in the central nervous system determines the type of visual deficit that occurs, which may or may not have significant visual consequences.9 As in this case, a left homonymous visual field defect results when a lesion affects the right visual cerebral cortex.
There are no standardized treatment protocols for primary central nervous system lymphoma secondary to PTLD.3,5,6 The most common treatments for DLBCL include corticosteroids for cerebral edema, whole-brain radiotherapy, and high-dose methotrexate.1,2,5,6 Rituximab may be beneficial as the medication targets specific antigens on B-cells.2,3,5,6 PTLD patients with monomorphic lymphoma have a median survival rate of nine years, with age 50 years and older being a significant risk factor in prognosis.2 As demonstrated in our case, prompt and ongoing medical intervention through a multidisciplinary approach is necessary for PTLD patients. In addition to the primary treatment team, including neurosurgery and oncology, optometrists and blind rehabilitation specialists should provide appropriate vision rehabilitation services when the visual system is affected, with the goal of maximizing the quality of life of the patient.
CONCLUSION
Post-transplant lymphoproliferative disorder rarely affects the central nervous system although it can have visually significant consequences depending on the location of intracranial involvement. Patients need to undergo routine and supplemental optometric testing and consider an expeditious referral to the next level of care when neurological findings are noted on examination. Ocular rehabilitation techniques can also improve the quality of life in patients with acquired brain injury. A multidisciplinary care team is needed to meet the patient’s needs, as no standardized treatment protocols exist.
REFERENCES
- Horvat M, Zadnik V, Setina T et al. Diffuse large B-cell lymphoma: 10 years’ real-world clinical experience with rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisolone. Oncology Letters 2018; 15: 3602-3609.
- Major A, Kamdar M. Management of non-diffuse large B-cell lymphoma post transplant lymphoproliferative disorder. Curr Treat Options in Oncol 2018; 19-33.
- Xu H, Rewerska J, Aardsma N et al. EBV-positive post-transplant lymphoproliferative disorder presenting as primary diffuse large B-cell lymphoma of the central nervous system. Folia neuropathol 2017; 55: 221-226.
- Zimmermann H, Ulrich Trappe R. Therapeutic options in post-transplant lymphoproliferative disorders. Ther Adv Hematol 2011; 2(6): 393-407.
- Batchelor T, Thye L, Habermann T. Current management concepts: primary central nervous system lymphoma, natural killer T-cell lymphoma nasal type, and post-transplant. American Society of Clinical Oncology Educational Book 2016; 35: 354-366.
- Kaulen L, Galluzzo D, Hui P et al. Prognostic markers for immunodeficiency‑associated primary central nervous system lymphoma. J Neurooncol 2019; 144: 107-115.
- Cavaliere R, Petroni G, Lopes M et al. Primary central nervous system post-transplantation lymphoproliferative disorder. Cancer 2010; 116(4): 863-870.
- Smatti M, Yassine H, AbuOdeh A et al. Prevalence and molecular profiling of Epstein Barr virus (EBV) among healthy blood donors from different nationalities in Qatar. PLoS ONE 2017; 12: e0189033.
- Ropper A, Samuels M, Klein J et al. Adam’s and Victor’s Principles in Neurology. 11th ed. New York: McGraw Hill Education, 2019. p 3.